In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).
Quick Summary
Polyclonal antibodies are generated by immunizing a host animal with a target antigen, triggering multiple B-cell clones to produce a heterogeneous mixture of immunoglobulins. This guide covers every step, from antigen design and adjuvant selection to immunization, serum harvest, purification, and quality validation with protocols, tables, and decision criteria researchers need to succeed.
Polyclonal antibodies (pAbs) are produced by injecting a specific antigen into a host animal, most commonly a rabbit or goat, which activates multiple B-lymphocyte clones, each secreting antibodies that bind distinct epitopes on that antigen. The resulting serum contains a heterogeneous mixture of high-affinity immunoglobulins that collectively recognize the antigen from multiple binding angles. This multi-epitope coverage makes polyclonal antibodies exceptionally sensitive and broadly applicable in Western blotting, ELISA, immunohistochemistry (IHC), and immunoprecipitation.
Understanding how polyclonal antibodies are produced, and where each decision point matters, is critical for obtaining high-titer, high-specificity antiserum that performs reliably across experimental formats.
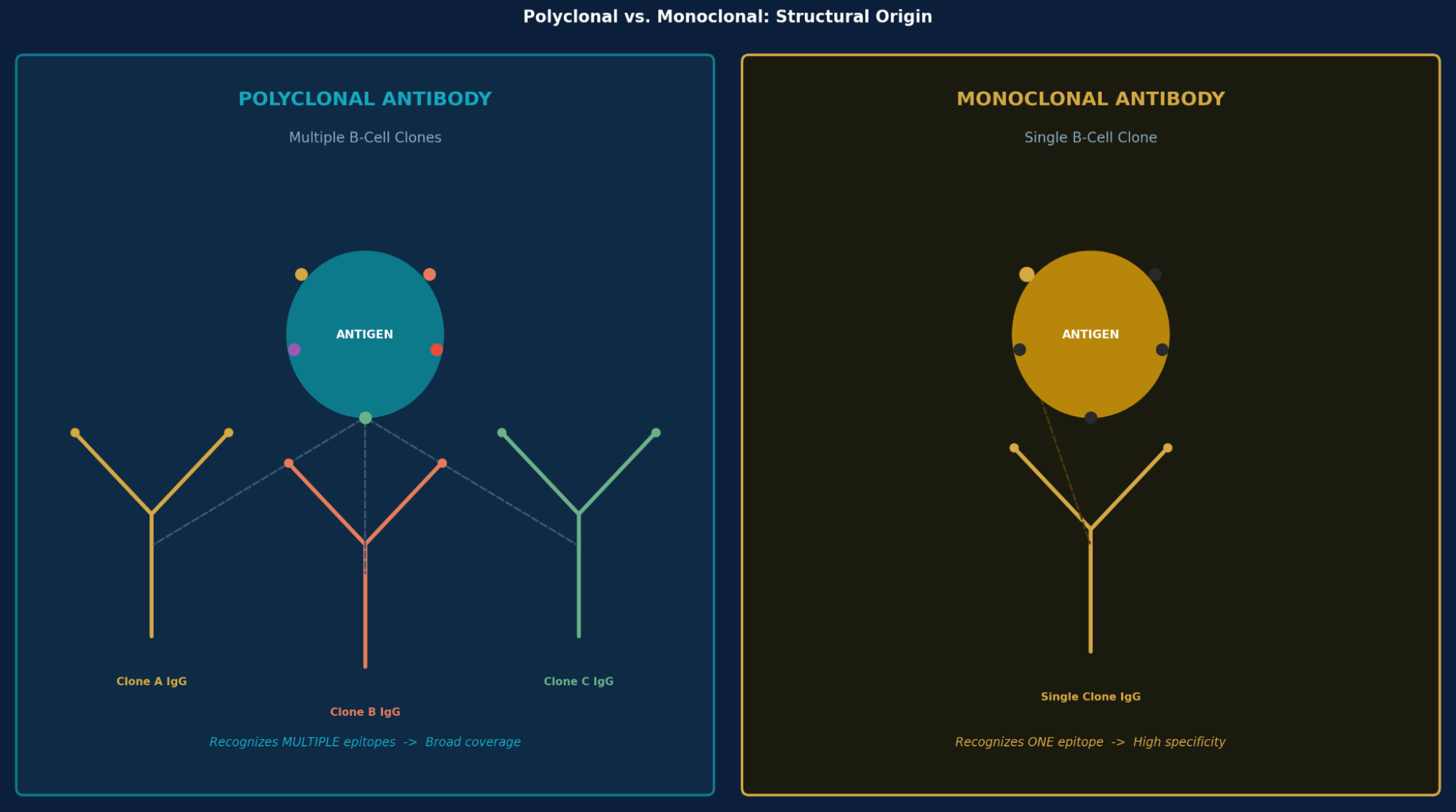
What Is a Polyclonal Antibody?
A polyclonal antibody is a mixture of immunoglobulins produced by multiple distinct B-lymphocyte clones, each targeting a different epitope on the same antigen. This contrasts with monoclonal antibodies, which originate from a single clone and recognize only one epitope.
Because polyclonal antibodies bind multiple epitopes simultaneously, they deliver stronger signal amplification in assays and can still detect antigen even after partial denaturation, a significant advantage in SDS-PAGE-based Western blotting.
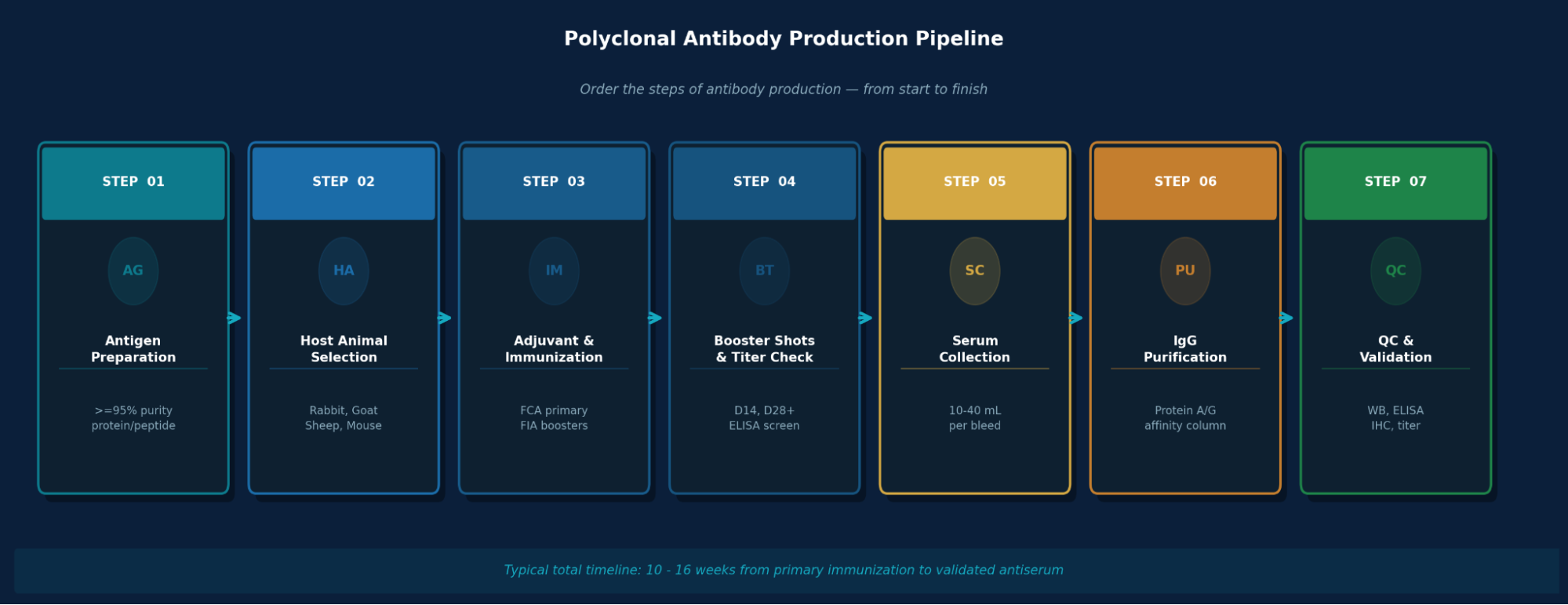
How Polyclonal Antibodies Are Produced: Step-by-Step
Here is the detailed explanation of PAb production:
Step 1: Antigen Preparation
Antigen quality is the single most critical variable in polyclonal production. Even trace impurities (<1%) with strong immunogenicity can introduce off-target antibody populations that contaminate the final antiserum.
- Protein antigens: Recombinant proteins expressed in E. coli, CHO, or HEK293 cells; purity ≥95% (SDS-PAGE confirmed).
- Peptide antigens: Short peptides (<15 kDa) must be conjugated to a carrier protein - Keyhole Limpet Hemocyanin (KLH), BSA, or thyroglobulin — to become immunogenic.
- Native proteins: Tissue-purified antigens retain conformation but carry contamination risks.
- Antigen should be sterile-filtered (0.22 µm), free of SDS, urea, acetic acid, and endotoxins.
⚠ Key Consideration:Peptides larger than 25–30 kDa typically do not require carrier conjugation. For smaller peptides, KLH conjugation is the most immunogenic choice due to KLH's high molecular weight (~4,500–13,000 kDa) and multiple surface lysines for coupling.
Step 2: Host Animal Selection
The host animal determines serum volume, antibody diversity, and production timeline. Rabbits are the standard for most research-grade polyclonal antibody generation, but the decision depends on downstream volume needs and species cross-reactivity requirements.
| Host Animal | Serum Yield / Bleed | Production Scale | Key Advantages | Limitations |
|---|---|---|---|---|
| Rabbit (Most Common) | 25–40 mL | Research / mid-scale | High titer, easy handling, well-characterized IgG | Limited volume per animal |
| Goat | 200–400 mL | Large-scale / diagnostic | Very high volume, commonly used for secondary antibodies | Longer immunization cycles |
| Sheep | 200–300 mL | Large-scale / diagnostic | Robust immune response, less prone to cross-reactivity with human IgG | Requires more antigen per dose |
| Mouse | 0.5–1 mL | Small-scale / scarce antigen | Genetic tools available; useful for MAb precursor screening | Very low yield per animal |
| Guinea Pig | 3–5 mL | Complement / research | Unique epitope specificity; natural complement source | Limited supplier protocols |
NOTE: Using 2–3 animals per antigen is recommended to account for individual immune response variability and ensure sufficient antibody diversity.
Step 3: Adjuvant Selection & Immunization Protocol
Adjuvants enhance and prolong the immune response by creating a depot effect at the injection site, stimulating antigen-presenting cells, and inducing cytokine secretion. Adjuvant selection directly affects antibody titer and the inflammatory burden on the animal.
- Freund's Complete Adjuvant (FCA): Water-in-mineral-oil emulsion containing killed Mycobacterium tuberculosis; gold standard for primary immunization; produces the highest titers but causes localized granuloma.
- Freund's Incomplete Adjuvant (FIA): Same emulsion without mycobacteria; used exclusively for booster injections.
- TiterMax Gold™ / RIBI™ / Alum: Less inflammatory alternatives; preferred in protocols governed by 3R principles.
Standard rabbit immunization schedule (100 µg antigen/dose):D0: Pre-immune bleed + Primary ImmunizationCollect baseline serum. Inject antigen in FCA across 8–10 subcutaneous sites on the dorsal surface.D14: First BoosterSame antigen dose in FIA (subcutaneous or intradermal). Avoids inflammatory escalation of FCA.D21: Test Bleed — ELISA Titer ScreenCollect 5–10 mL via ear vein. ELISA OD reading determines response adequacy before further boosting.D28+: Additional Boosters (every 2–4 weeks)Continue antigen-in-FIA boosts, monitoring titer after each. Affinity maturation improves antibody quality with each cycle.
Step 4: Blood Collection & Serum Harvest
Once titers peak and stabilize (typically 6–12 weeks post-primary immunization), serum collection begins. Strict volume limits apply: no more than 10–15% of total blood volume collected every two weeks to prevent anemia or hypovolemic shock.
- Survival bleeds (rabbit): Marginal ear vein or central auricular artery; yields 10–25 mL per collection.
- Terminal bleed: Cardiac puncture or carotid exsanguination performed under anesthesia; maximizes final volume.
- Whole blood is allowed to clot at room temperature (30–60 min) or 4°C, then centrifuged at 1,000–2,000 × g for 15 min to isolate clarified antiserum.
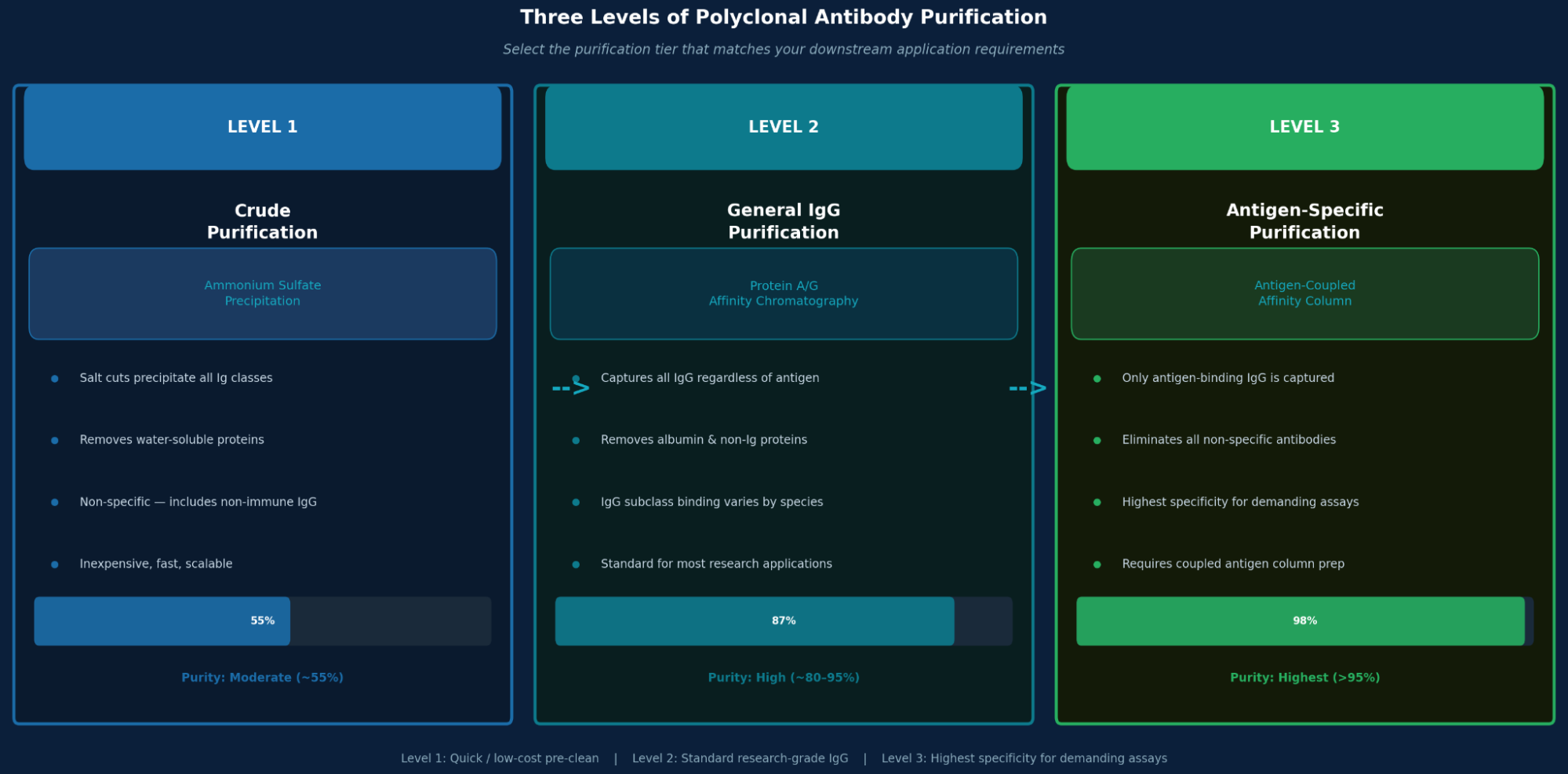
Step 5: Antibody Purification
Step 6: Quality Control & Validation
A purified polyclonal antibody must be validated before use. Validation data determines reliability and supports reproducibility in peer-reviewed research.
| QC Parameter | Method | Acceptance Criteria |
|---|---|---|
| Antibody Titer | ELISA (serial dilution) | Positive OD at ≥1:10,000 dilution (typical minimum) |
| Specificity | Western Blot | Single band at expected molecular weight |
| Cross-Reactivity | Multi-antigen ELISA panel | No significant binding to structurally related non-targets |
| Concentration | A280 absorbance (NanoDrop) | Reported as mg/mL; aliquot and store at –20°C or –80°C |
| Application Performance | WB, IHC, ELISA, IP | Passes in ≥1 validated application; cross-application validation preferred |
Key Applications of Polyclonal Antibodies in Research
- Western Blot (WB): Multi-epitope binding provides strong signal even with partially denatured antigen; ideal for detecting low-abundance proteins.
- ELISA: Used in both sandwich (capture + detection) and indirect formats; high sensitivity due to polyclonal coverage.
- Immunohistochemistry (IHC): Superior tissue signal amplification compared to monoclonals; performs well on FFPE sections.
- Immunoprecipitation (IP): Higher pull-down efficiency due to recognition of multiple surface epitopes on native antigen.
- Flow Cytometry (FC): Useful for surface antigen detection on live or fixed cell populations.
- Neutralization Assays: pAbs targeting multiple viral epitopes confer broad neutralization in virology research.
For validated, application-tested polyclonal antibodies across these formats, explore the AAA Biotech polyclonal antibody catalog.
Known Challenges in Polyclonal Antibody Development
- Batch-to-Batch Variability: Each immunization cycle produces a different antibody population; titers, affinities, and epitope distributions shift between bleeds and animals.
- Cross-Reactivity: Impurities in antigen preparation elicit off-target antibodies that increase assay background, especially in IHC and Western blot.
- Supply Finitude: Production is limited by the host animal's lifespan and bleeding capacity; once an animal is exhausted, the exact antibody mixture cannot be identically reproduced.
- Ethical Considerations: Repeated immunization and bleeding imposes physiological burden; 3R principles (Replacement, Reduction, Refinement) must be followed in all institutional animal protocols.
- Non-Specific Immune Responses: Animals may carry pre-existing antibodies to environmental antigens that cross-react with target epitopes; always collect and bank pre-immune serum as a control.
Final Thoughts
Polyclonal antibody production remains a cornerstone of biomedical research, valued for its speed, sensitivity, and multi-epitope coverage. Successful polyclonal antibody generation depends on disciplined antigen preparation, deliberate host animal and adjuvant selection, a tightly monitored immunization schedule, and rigorous post-harvest validation. Each step is interconnected: failures in antigen purity propagate through every subsequent stage. Researchers who invest in protocol optimization upfront consistently obtain high-titer, specific, and reproducible antiserum that delivers across multiple assay platforms.
For access to rigorously validated polyclonal antibodies, ELISA kits, and recombinant proteins for your research workflows, visit the AAA Biotech catalog.
Faq's
What is a polyclonal antibody and how does it differ from a monoclonal antibody?
A polyclonal antibody is a mixture of immunoglobulins produced by multiple B-cell clones, each binding a different epitope on the same antigen. A monoclonal antibody originates from a single clone and targets only one epitope. Polyclonal antibodies offer broader antigen coverage and higher sensitivity; monoclonal antibodies offer higher specificity and consistent batch-to-batch performance.
How long does polyclonal antibody production take from start to finish?
From primary immunization to validated antiserum, the full polyclonal antibody production timeline in rabbits is typically 10–16 weeks. This includes antigen preparation (~1–2 weeks), primary immunization and first booster (~4 weeks), titer monitoring and additional boosts (~4–8 weeks), and purification/QC (~2 weeks). Longer timelines apply when titer responses are slow or additional boosters are needed.
Which host animal produces the highest titer polyclonal antibodies?
Rabbits consistently produce the highest titer polyclonal antibodies for most antigens relative to their body size, with serum yields of 25–40 mL per bleed. Goats and sheep produce far greater volumes (200–400 mL) but are primarily used when large-scale production is required, such as for generating secondary antibodies used in laboratory kits.
Why is antigen purity so critical in polyclonal antibody generation?
Impure antigens cause the host animal's immune system to generate antibodies against contaminants alongside the target. Even <1% impurities with strong immunogenicity can significantly pollute the final antibody pool with off-target specificities, increasing background noise in assays like IHC, ELISA, and Western blot. Antigen purity ≥95% (SDS-PAGE confirmed) is the minimum accepted standard.
What are the main quality control tests required after polyclonal antibody purification?
Standard QC for purified polyclonal antibodies includes: ELISA titer determination (to confirm sufficient antigen-binding activity), Western blot specificity testing (to verify the antibody detects the correct band), cross-reactivity screening (against related antigens), concentration measurement by A280, and application-specific validation (WB, IHC, ELISA, or IP depending on intended use).

 Cynthia
Cynthia