In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

Key Takeaway
- Antibody validation ensures results are accurate and reproducible
- Poor validation leads to misleading and non-reproducible data
- Multiple strategies confirm specificity in different ways
- Validation must match your specific application and conditions
- Lot-to-lot variability affects consistency in polyclonal antibodies
- Evaluating supplier data carefully helps avoid experimental failure
Every researcher has been there. An experiment fails. The protocol is correct. The cells look healthy. But the results make no sense and cannot be reproduced. More often than not, the antibody is the problem.
Antibodies are among the most widely used tools in life science research, and also among the most poorly validated. Studies estimate that irreproducible research costs the US $28 billion annually, with biological reagents identified as a leading cause.
The word "validated" on a product page sounds reassuring. But it covers a wide range of actual testing, from a simple binding check to rigorous multi-method characterization. Most researchers only discover this gap after something goes wrong.
Understanding what proper validation of antibodies actually involves is the first step toward building experiments that produce results you can trust and reproduce consistently.
What Does Validation of Antibodies Actually Mean?
Antibody validation is the process of experimentally confirming that an antibody detects its intended target, and only that target, under the exact conditions of your experiment. It is not a single test. It is a structured process that combines multiple strategies to build confidence in an antibody's specificity and reliability.
Most suppliers perform basic quality control testing, which confirms purity and antigen binding in a controlled format. This is not the same as scientific validation. Scientific validation goes further by confirming specificity in the actual application, using biologically appropriate positive and negative controls in a system that reflects real experimental conditions.
These are not minor differences in degree. They are fundamentally different levels of evidence, and treating them as equivalent is one of the most common mistakes researchers make when selecting reagents.
Did You Know?
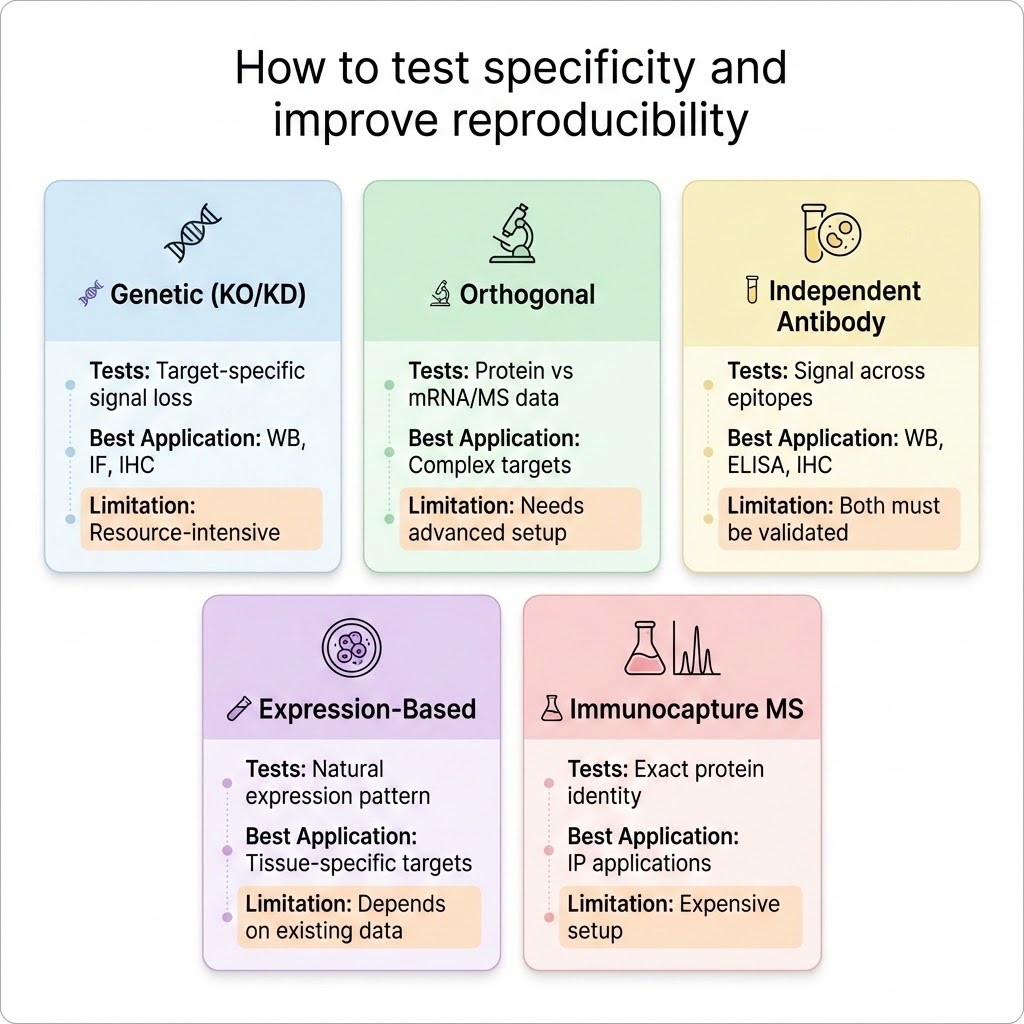
The International Working Group for Antibody Validation (IWGAV) published a five-strategy framework in Nature Methods (2016) that remains the most rigorous and widely accepted standard in the field. Each strategy tests a different dimension of antibody specificity testing, and together they provide a complete picture of whether an antibody is genuinely fit for purpose.
Antibody Validation Methods: What Each Strategy Tests
Not all validation approaches test the same thing. Different targets, applications, and research contexts call for different methods, and each one reveals a distinct aspect of antibody performance.
Knowing what each strategy actually confirms, and where its limits are, is what allows researchers to evaluate supplier data with confidence rather than assumption.
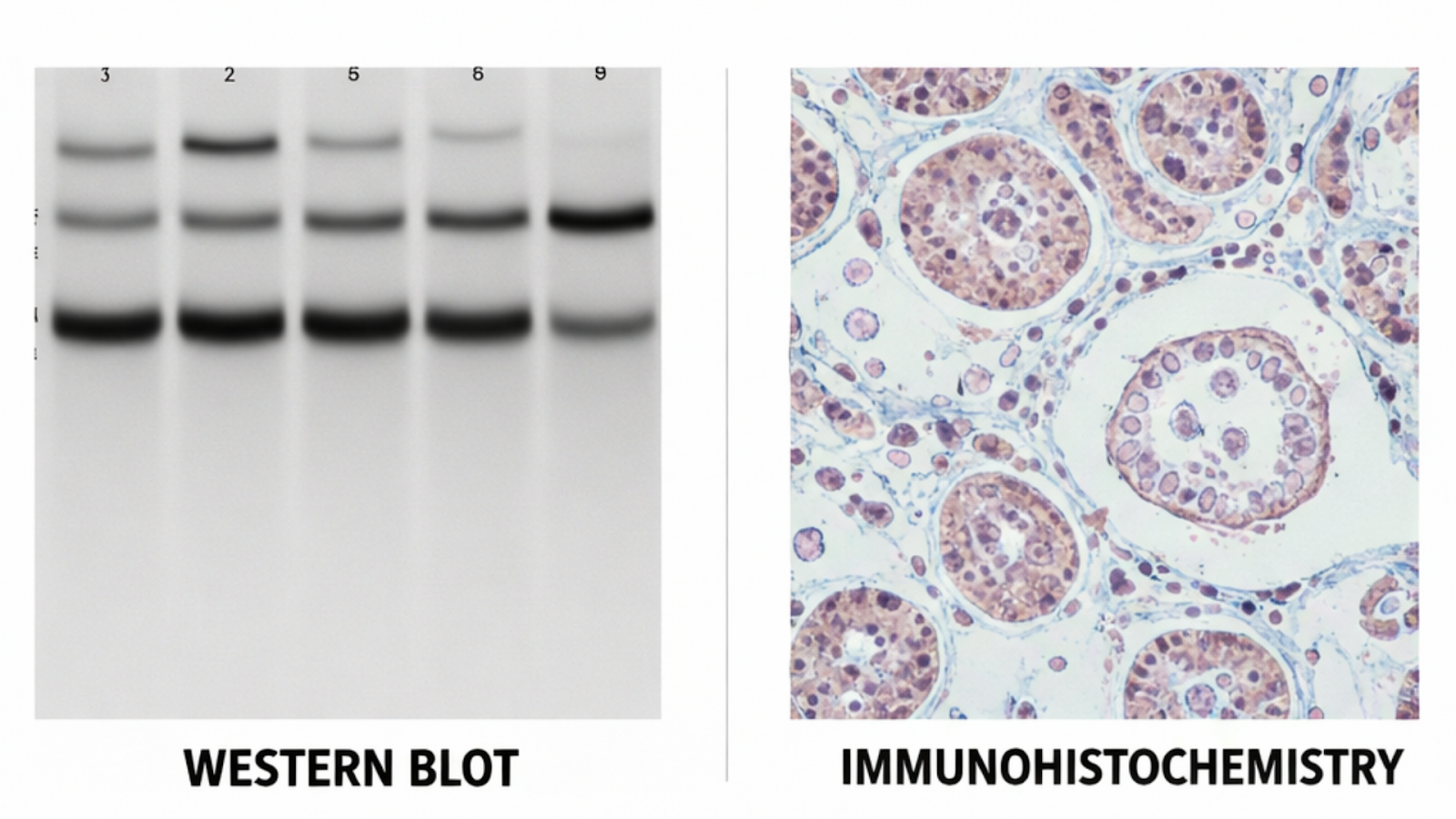
Genetic Validation (Knockout and Knockdown Controls)
- What it involves: Testing antibody performance in cell lines where the target gene has been eliminated by CRISPR knockout or suppressed by siRNA knockdown.
- What it confirms: If the signal disappears or significantly reduces in the knockout or knockdown lane, the antibody is detecting the intended target with strong specificity.
- Practical reality: This is considered the most rigorous single approach for Western blot and immunofluorescence. Its presence in a supplier's validation data is a meaningful quality signal precisely because it is inexpensive and relatively difficulty to produce.
- What it cannot tell you: Performance in other applications or other species is not confirmed by knockout validation alone.
Orthogonal Validation
- What it involves: Comparing antibody-based protein detection against a completely independent, non-antibody method, such as mass spectrometry or mRNA expression correlation.
- What it confirms: The protein that the antibody detects corresponds to what the gene is actually expressing in that biological system, ruling out the possibility that the signal is an antibody artefact unrelated to true protein expression.
- Practical reality: Particularly valuable for novel or poorly characterized targets where expression data is limited, and cross-reactivity risk is higher.
- What it cannot tell you: It does not confirm performance across all experimental applications or verify detection under denaturing or fixation conditions.
Independent Antibody Validation
- What it involves: Using two or more antibodies that recognize different epitopes on the same target protein and confirming that both produce concordant results.
- What it confirms: The detected signal reflects genuine target expression rather than an artefact of one particular antibody's binding characteristics.
- Practical reality: This approach is accessible to most labs without specialist infrastructure and does not require knockout cell lines or mass spectrometry equipment.
- What it cannot tell you: If both antibodies are poorly characterized, concordant results do not equal validated results. Each antibody must itself carry independent evidence of specificity for this strategy to be meaningful.
Expression-Based Validation
- What it involves: Confirming the antibody detects a signal in cell lines or tissues known to express the target and produces no signal in those that do not, using reference data from databases such as the Human Protein Atlas.
- What it confirms: The antibody is targeting the correct protein in a biologically meaningful way, consistent with known expression patterns.
- Practical reality: Quick and widely applicable without specialist equipment. Particularly useful for tissue-specific targets where expression patterns are well established.
- What it cannot tell you: For novel targets with limited prior expression data, this strategy loses reliability. If the reference data is incomplete, the validation conclusion will be too.
Immunocapture Mass Spectrometry
- What it involves: Using the antibody to immunoprecipitate the target protein from a complex cell lysate, then confirming the identity of the captured protein by mass spectrometry.
- What it confirms: The protein being pulled down is definitely the intended target, making this the highest confidence identification method across all five strategies.
- Practical reality: Gold standard for immunoprecipitation applications. Expensive and technically demanding, it is typically performed by specialist core facilities rather than individual research labs.
- What it cannot tell you: It does not confirm performance in other applications. Successful immunocapture confirms binding under native conditions but says nothing about antibody performance when the antigen is denatured or chemically fixed.
What Are All Five Validation Strategies Actually Testing?
Regardless of which strategy is applied, every validation method is ultimately testing the same thing: antibody specificity. An antibody that binds something other than its intended target is not merely unreliable - it is actively misleading, and produces data that looks valid but cannot be reproduced because the signal was never “real”.
Why Application-Specific Validation Is Non-Negotiable
An antibody validated for Western blot has been confirmed to detect a denatured, linearly presented protein epitope under SDS-PAGE conditions. That confirmation tells you almost nothing about how it will perform in immunohistochemistry, where the antigen is fixed in tissue and conformational epitopes may be partially or fully altered by fixation chemistry.
| Application | Antigen Condition | Key Validation Requirement |
|---|---|---|
| Western Blot | Denatured, linear epitope | Signal in positive control, loss in KO lane |
| IHC / IF | Fixed tissue, antigen retrieval required | Correct localization, negative tissue control |
| ELISA | Solution-phase, native conformation | Sensitivity, dynamic range, and no cross-reactivity |
| Flow Cytometry | Intact cells, surface or intracellular | Correct population gating, isotype control |
| Immunoprecipitation | Native protein, complex mixture | Clean pull-down confirmed by secondary method |
Always confirm that the antibody has been validated specifically for your application, in your target species, and in a biologically comparable system. Predicted reactivity is not validated reactivity.
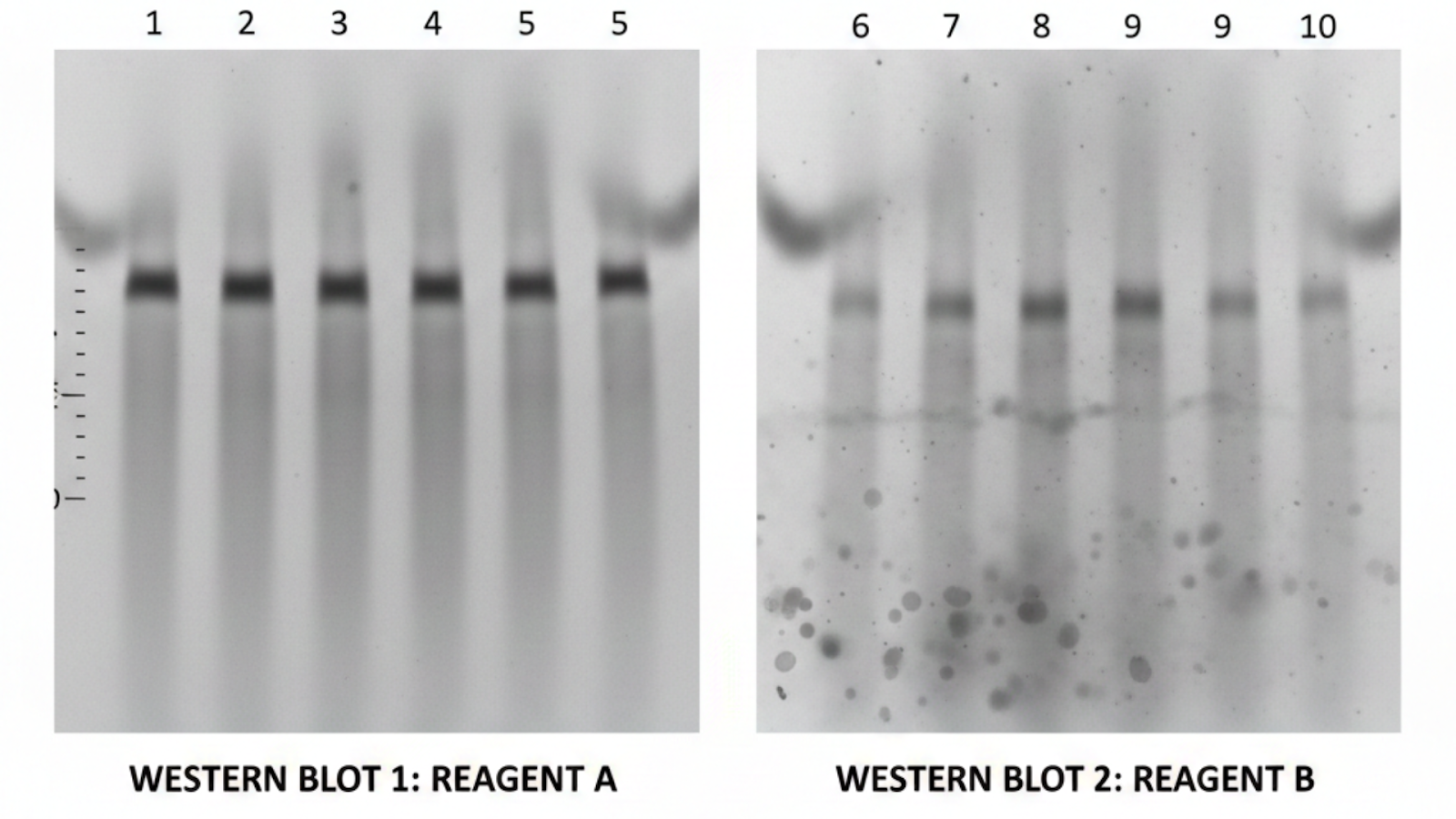
Lot-to-Lot Variability: The Silent Reproducibility Killer
Why does the same catalog number sometimes produce different results six months apart?
Polyclonal antibodies come from animal immunizations. Each new lot is a biologically distinct mixture of antibody clones with different binding profiles. The catalog number stays the same. The antibody does not.
This is not a quality control failure. It is simply how polyclonal antibodies work. And it is a variable most researchers do not account for until an experiment fails to replicate.
"Lot-to-lot variability is not a quality control failure; it is a structural property of polyclonal antibody biology. The solution is not to demand better batches. It is to choose a format where batches are irrelevant."
Recombinant monoclonal antibodies are produced from a cloned, sequenced gene in a stable cell line. The same molecule is delivered every time, with no biological variation between lots. For any assay that is repeated, published, or shared across a lab, this format removes one of the most common and least visible sources of irreproducibility.
How to Validate Antibodies: Evaluating Supplier Data Before You Buy
Not all product pages are equal. Knowing what to look for and what to be cautious about is the most practical step a researcher can take before committing to a new antibody.
Green flags on a product page:
| Green Flag | Why It Matters |
|---|---|
| Knockout or knockdown validation images | Confirms target-specific signal with the highest rigor |
| Application-specific testing matching your technique | Directly relevant to your experimental system |
| Species reactivity confirmed experimentally | Predicted reactivity is not validated reactivity |
| Recombinant monoclonal format stated | Eliminates lot-to-lot variability structurally |
| Publication citations in comparable systems | Independent, peer-reviewed validation |
Red flags on a product page:
| Red Flag | What It Signals |
|---|---|
| "Predicted to react" without experimental data | Computational estimate, not biological confirmation |
| Validation images only in one application for a multi-application listing | Untested performance outside the listed application |
| No knockout or orthogonal validation data | Basic QC only — not scientific validation |
| No information on antibody format or clonality | Cannot assess lot-to-lot risk |
| Vague species reactivity with no experimental images | Cross-reactivity is assumed, not confirmed |
Final Thought
Reproducibility does not happen by luck. It comes down to the decisions you make before the experiment even starts, and choosing the right validated antibody is one of the most important ones.
Journals are asking for validation data. Funding bodies are paying closer attention to reagent quality. And well-validated recombinant antibodies are now easier to access and more affordable than ever.
The researchers who get consistent, publishable results are not doing something magical. They are just making better reagent choices. Browse the AAA Bio fully validated antibody catalog and find the right antibody for your application.
Faq's
What is the difference between antibody validation and quality control testing?
Quality control testing confirms basic properties like purity and binding under controlled conditions. Antibody validation goes further, proving specificity and performance in real experimental settings using biological controls. Validation ensures reliable, reproducible results, while quality control only verifies manufacturing consistency.
How to validate antibody specificity in my own lab before committing to a long-term study?
Start with a pilot experiment using known positive and negative samples. Include controls such as knockout or knockdown if available, verify expected band size or localization, and test across your intended application to confirm a consistent, target-specific signal before scaling up.
If an antibody is validated for Western blot, can I use it for immunohistochemistry without re-validating?
No. Western blot validation confirms performance under denatured conditions, while immunohistochemistry involves fixed tissues and altered epitopes. Each application requires separate validation to ensure accurate binding and localization under its specific experimental conditions.
Why do recombinant monoclonal antibodies offer better experimental reproducibility than polyclonal antibodies?
Recombinant monoclonal antibodies are produced from a defined genetic sequence, ensuring consistent structure and performance across batches. In contrast, polyclonal antibodies vary between lots, leading to inconsistent binding profiles and reduced reproducibility in long-term or comparative experiments.

 Cynthia
Cynthia