In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

Key Takeaway
- High protein expression doesn’t guarantee activity.
- Misfolding and inclusion bodies reduce functional yield.
- Codon optimization and vector design improve translation.
- Host selection affects folding and post-translational modifications.
- Slow induction and lower temperatures boost solubility.
- Fusion tags and chaperones enhance active protein yield.
- Optimized culture conditions maximize productivity.
- A checklist ensures reproducible, functional protein results.
Have you ever expressed a protein successfully but ended up with insoluble or inactive results?
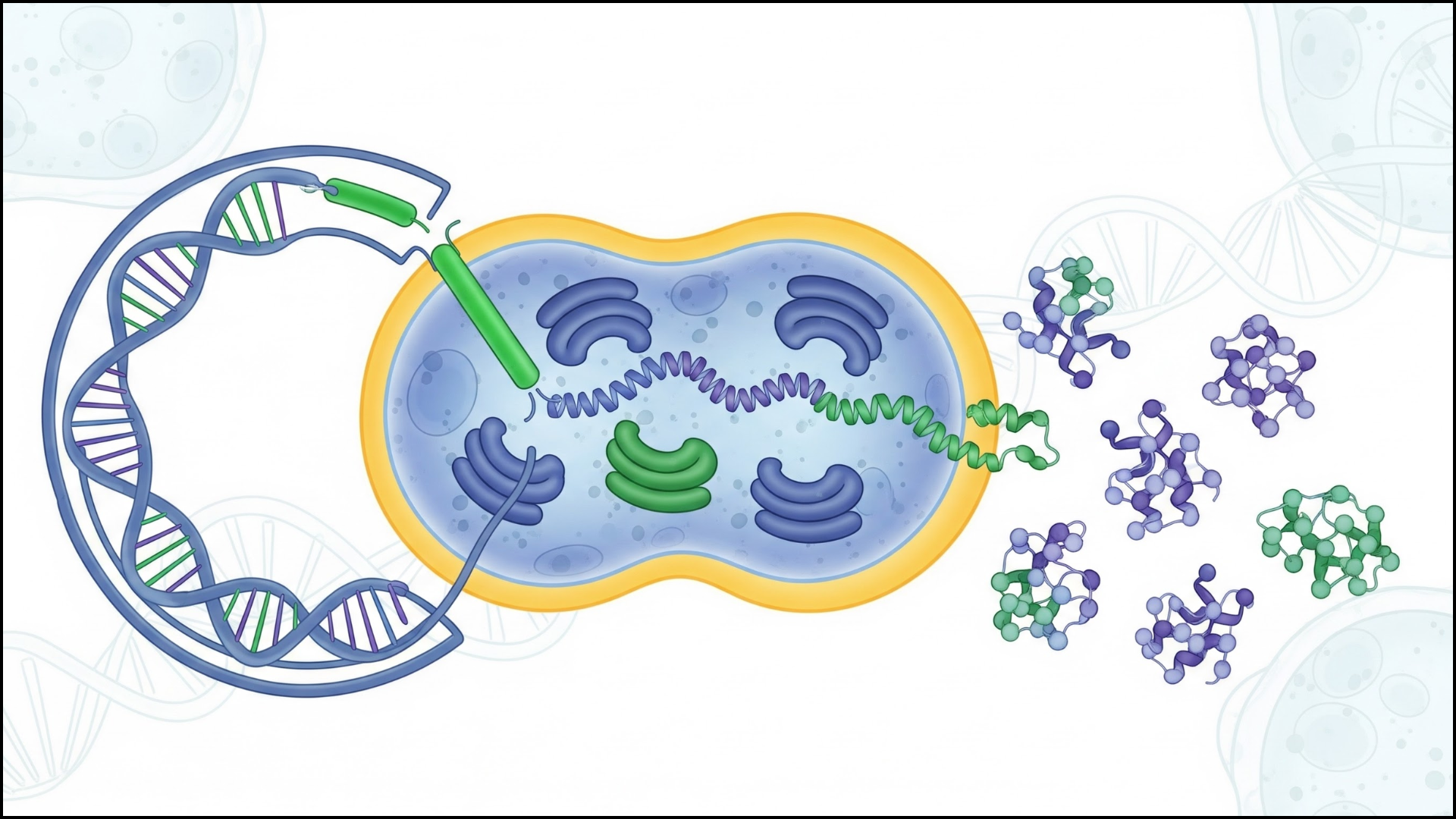
It’s a common challenge in recombinant protein research. High expression levels do not always produce functional proteins. Many recombinant proteins misfold or aggregate during expression.
In fact, studies show that nearly 70% of recombinant proteins expressed in bacterial systems, such as E. coli, can form insoluble inclusion bodies when expression conditions are not optimized.
As the global protein expression market is expected to exceed $6 billion by 2032, researchers are focusing more on producing active protein, not just high quantities. So how can you improve functional protein yield?
The answer lies in optimizing several key factors. These include codon usage, host selection, induction conditions, fusion tags, and culture parameters. This guide covers practical protein expression techniques that improve protein folding and activity. It also explains how to achieve better protein expression and purification results.
Why Active Protein Yield Matters More Than Total Protein
In recombinant protein research, quantity alone does not guarantee success. What truly matters is whether the produced protein retains its proper structure and biological function.
When High Expression Creates Problems
Many expression systems are designed to produce proteins very quickly. But when protein synthesis happens too fast, the cell’s folding machinery cannot keep up. As a result, newly produced proteins may misfold and accumulate as insoluble inclusion bodies. These aggregates usually lack biological activity and can be difficult to recover during purification.
Why Functional Protein Is the Real Goal
For most researchers, the real objective is not simply producing more protein but obtaining a stable and functional active protein. The protein should be properly folded, biologically active, and stable throughout purification and analysis.
The demand for functional proteins is increasing in biotechnology and pharmaceutical research. Protein-based therapeutics are now one of the fastest-growing drug classes. Market studies estimate the therapeutic protein market will grow at a CAGR of around 8.5% from 2020 to 2027.
This trend increases the demand for reliable production of active protein.
Understanding the Key Factors That Influence Protein Expression
Producing a protein in the lab may seem straightforward. Insert the gene, grow the cells, and induce expression. But in reality, many proteins fail to fold properly or lose their biological activity.
That happens because protein production depends on several biological variables working together. Factors such as codon usage, host system, expression speed, and temperature all influence whether the final product becomes a functional active protein.
Understanding these factors is the first step toward improving your protein expression methods and achieving reliable results.
| Factor | Why It Matters |
|---|---|
| Codon usage | Controls translation efficiency |
| Host system | Determines folding and post-translational modifications |
| Expression rate | Too fast leads to aggregation |
| Temperature | Influences protein folding |
| Culture conditions | Affect cell health and productivity |
How Can the Right Genetic Design Improve Protein Expression?
Have you ever cloned and expressed a protein successfully, but later discovered that most of it was inactive or stuck in inclusion bodies? This is a common challenge in recombinant protein research. High expression levels do not always translate into functional proteins.
Often, the solution starts much earlier than the expression step. Careful gene design and proper protein optimization can greatly improve both protein yield and biological activity.
Codon Optimization for Efficient Translation
Different organisms prefer certain codons to produce proteins. When a gene contains rare codons for the host organism, the ribosome may slow down or pause during translation. This can lead to incomplete proteins or incorrect folding. Adjusting the gene sequence to match the host’s preferred codons helps improve translation efficiency and protein yield.
Vector Design and Promoter Selection
Expression vectors control how strongly a protein is produced. Strong promoters like T7 drive high expression, but excessive production can overwhelm the cell’s folding machinery. Using inducible systems and appropriate plasmid copy numbers helps balance expression speed with proper protein folding.
How Do You Choose the Right Host System for Protein Expression?
One of the first questions researchers ask is: Which expression system will give me a functional protein?
The answer depends on the protein’s complexity and folding requirements. Choosing the right host early can save time and improve the success of protein expression and purification.
- Bacterial Expression Systems (E. coli)
E. coli is a popular starting point for recombinant protein experiments.
Advantages
- Grows very quickly
- Affordable for large experiments
- Produces high protein yields
Limitations
- Cannot perform many post-translational modifications
- Proteins may form insoluble aggregates
- Yeast Expression Systems
Yeast systems provide some advantages of eukaryotic cells while remaining relatively easy to grow.
Benefits
- Improved folding compared to bacterial systems
- Ability to secrete proteins into the culture medium
- Mammalian Expression Systems
Mammalian cells are preferred when protein structure and function must closely resemble human proteins.
Examples
- HEK293
- CHO cells
Advantages
- Accurate glycosylation
- Ideal for antibodies and therapeutic proteins
What’s the Best Way to Induce Protein Expression Without Losing Activity?
Induction is where most proteins go wrong. The goal: slow and steady wins the race.
Tip 1: Keep It Cool
Lowering the temperature to 15–25°C slows protein synthesis.
Result: less aggregation, more active protein.
| Temperature | Outcome |
|---|---|
| 37°C | Fast expression, more aggregation |
| 18–25°C | Slow, soluble, active protein |
Tip 2: Optimize Inducer Concentration
Less can be more. Instead of the standard 1 mM IPTG, try 0.05–0.1 mM to give cells time to produce properly folded protein.
Tip 3: Follow a Simple Stepwise Induction Protocol
- Grow to OD600 0.4–0.8
- Add inducer
- Reduce temperature
- Express longer
Can Fusion Tags and Chaperones Boost Your Protein’s Solubility?
Proteins that misfold or aggregate can be a major obstacle in recombinant expression. Fusion tags and molecular chaperones act as helpers, keeping your protein soluble and functional. Using them is one of the most reliable protein expression techniques for challenging targets.
| Tag | Full Form | Benefit / How It Helps |
|---|---|---|
| MBP | Maltose Binding Protein | Acts as a “solubility booster,” keeping aggregation-prone proteins folded and active. Useful for difficult proteins. |
| GST | Glutathione S-Transferase | Improves folding efficiency and reduces misfolded or inactive proteins. Can also be used for affinity purification. |
| SUMO | Small Ubiquitin-like Modifier | Enhances folding and allows clean removal, leaving the native protein sequence intact. |
| His-tag | Polyhistidine Tag | Small tag that enables fast purification using affinity resins with minimal impact on folding. |
Quick Tip: For proteins that are hard to express, pairing fusion tags with chaperones can improve solubility and functional yield, making your protein expression techniques more effective.
How Can You Optimize Culture Conditions for Maximum Protein Yield?
Maximizing protein yield isn’t magic; it’s about optimal culture conditions.
- Feed Your Cells Right: Rich media like Terrific Broth or auto-induction give them the energy to make more protein.
- Right Time to induce: OD600 between 0.5–0.8 is the sweet spot for active, metabolically healthy cells.
- Keep an Eye on Oxygen & pH: Especially in bioreactors, these small tweaks maintain productivity.
Following these steps makes upscaling protein production from shake flasks to large bioreactors much easier.
Troubleshooting Common Protein Expression Problems
When protein expression fails, don’t panic. Applying proper protein expression methods can quickly turn things around.
| Problem | Solution | Explanation |
|---|---|---|
| No expression | Check for mutations or frameshifts in the plasmid | Sometimes the gene sequence has errors, or the plasmid has structural problems. Verifying the DNA ensures your cells have the correct template to produce protein. |
| Inclusion bodies | Reduce expression speed by lowering the temperature | Fast protein synthesis can overwhelm folding machinery, causing aggregation. Slowing translation at 15–25°C allows proper folding and improves solubility. |
| Protein degradation | Switch to protease-deficient hosts | Some host strains have proteases that chew up your protein. Using strains lacking these proteases, or adding inhibitors, keeps your protein intact. |
| Low activity | Test an alternative host system (E. coli → HEK293) | If the protein isn’t functional, it may need post-translational modifications. Switching to a mammalian host can produce an active protein with correct folding. |
Checklist for High-Quality Active Protein
Getting active protein doesn’t have to be guesswork. Make sure you’ve:
- Optimized your codons for smooth translation
- Picked the right host system
- Reduced induction temperature to give proteins time to fold
- Added fusion tags to improve solubility
- Adjusted the inducer concentration carefully
- Monitored media, pH, and oxygen levels
- Validated conditions for scale-up
Follow this checklist and watch your active protein yield improve!
Final Thought
Producing active protein is a balancing act. You need the right gene design, a suitable host, and carefully controlled culture conditions. Get these right, and you boost yield, activity, and reproducibility. It’s not just about quantity; it’s about quality.
If you are looking for proteins or reagents for your experiment, always choose high-quality options to ensure reliable results. AAA Bio provides the essential tools your lab needs, from proteins to reagents, to boost efficiency and accuracy in research.
“High protein expression isn’t winning by volume; it’s winning by function.”
Faq's
When should I use custom protein expression services?
Use them for difficult proteins—membrane proteins, toxic proteins, complex eukaryotic proteins, or therapeutic antibodies. Custom protein expression provides optimized hosts, tailored induction, and purification strategies to ensure high-quality, functional proteins quickly.
How do I increase the solubility of recombinant proteins?
Use fusion tags like SUMO or MBP and co-express with molecular chaperones like GroEL-GroES.
What is the optimal IPTG concentration?
Optimal IPTG varies by protein and host, but lower concentrations (0.05–0.1 mM) often work best. They slow expression, reduce aggregation, and improve soluble, active protein yield compared to standard 1 mM induction. Test small ranges for your protein.
What is the best temperature for protein expression induction?
For most E. coli systems, inducing at 16–20°C overnight is optimal for solubility.
How to optimize expression vectors for recombinant proteins?
Choose a strong but controllable promoter, select a compatible replication origin, and optimize codons for your host. Add solubility-enhancing tags if needed. Balancing expression strength with folding capacity ensures higher yields of active, functional protein.

 Cynthia
Cynthia