In this Article
- 1. Start With the Expression System: It Shapes Everything Downstream
- 2. Purity Grade: Align Specification With Experimental Need
- 3. Bioactivity: The Metric That Predicts Real-World Performance
- 4. Endotoxin Levels: The Variable That Quietly Ruins Experiments
- 5. Protein Tags: Useful Handles That Can Also Interfere
- 6. Formulation, Storage Conditions, and Stability
- 7. Species Compatibility and Cross-Reactivity
- 8. Supplier Reliability: Batch Consistency and Documentation
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).
Key Takeaways
- The expression system matters first. It determines folding, post-translational modifications, and downstream bioactivity.
- Purity grade must match your assay. Not every experiment demands >95%, but sensitive cell-based assays usually do.
- Bioactivity, not purity alone, predicts performance. Always check functional validation data on the COA.
- Endotoxin contamination is a hidden variable. Even trace levels can skew cell culture and in vivo results dramatically.
- Batch-to-batch consistency wins in the long term. Reproducible science requires a supplier you can verify and trust.
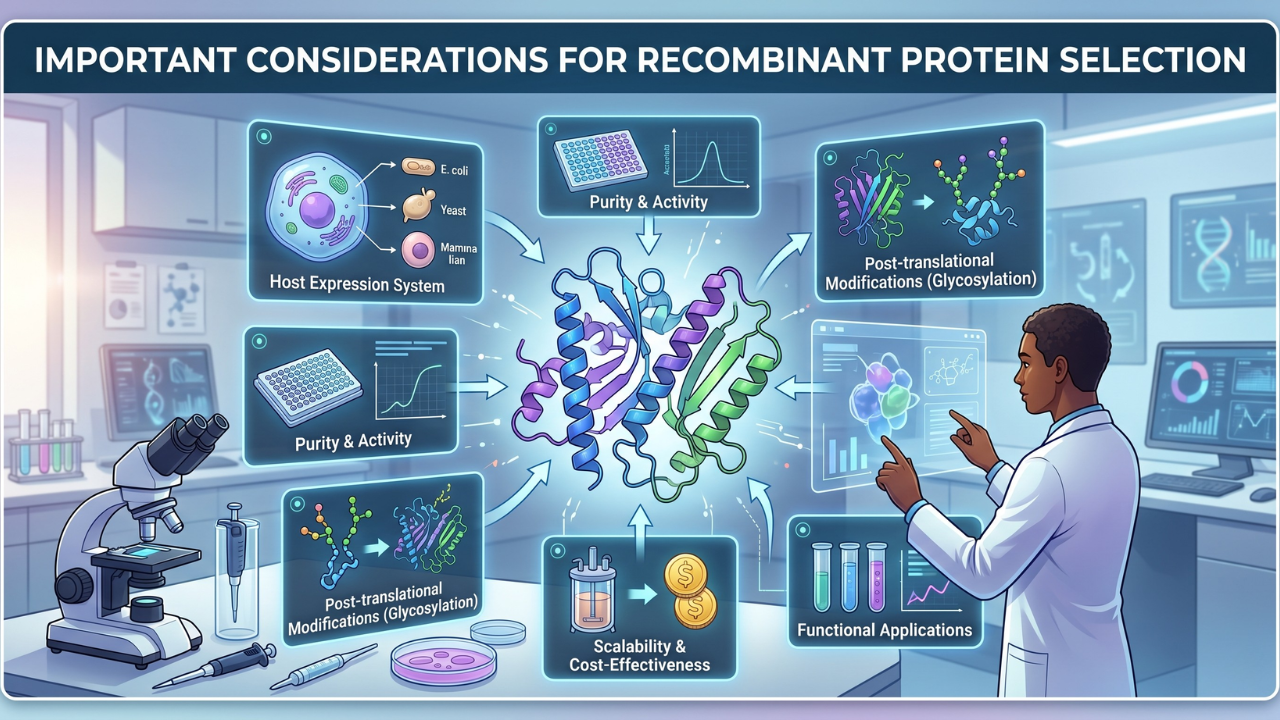
Selecting the right recombinant protein is one of the most consequential decisions a researcher makes before an experiment even begins, and yet it rarely receives the same scrutiny as downstream assay design.
Two different commercial recombinant protein vials labeled with identical names can produce entirely different outcomes depending on how the protein was made, how pure it is, whether it is biologically active, and whether it was stored correctly.
We will help you unpack every critical variable, so that your next decision, whether you are ordering recombinant cytokines for an immunology screen or a precise growth factor for stem cell differentiation, is grounded in science, not guesswork.
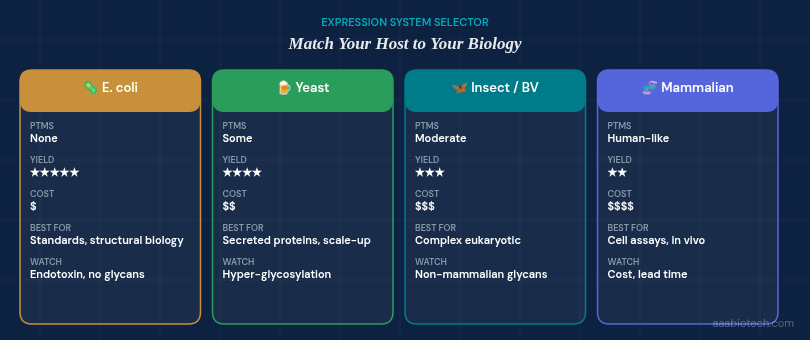
1. Start With the Expression System: It Shapes Everything Downstream
Before asking about purity or price, ask: where was this protein made?
The expression host controls folding, glycosylation, and post-translational modifications (PTMs), which, those three things together determine whether a protein behaves the way biology intended.
| Expression System | PTMs | Yield | Best Suited For | Watch Out For |
|---|---|---|---|---|
| E. coli (Bacterial) | None / Minimal | Very High | Structural studies, antibody production standards | Refolding issues, endotoxin |
| Mammalian (HEK293, CHO) | Human-like | Lower | Cell-based assays, therapeutic models | Higher cost, longer lead times |
| Insect (Sf9, Baculovirus) | Moderate | High | Complex eukaryotic proteins | Non-mammalian glycans can affect receptor binding |
| Yeast (Pichia, Saccharomyces) | Some | High | Secreted proteins, scalable production | Hypermannosylation |
For most cell culture work involving a recombinant growth factor — EGF, FGF, TGF-β, and related native proteins, mammalian expression is the gold standard because it preserves the glycosylation signatures that receptor-binding often depends on. However, many well-validated bacterial-produced proteins perform reliably when refolding and purification are tightly controlled.
The honest answer to which expression culture is right for you will always come back to one question: Does your specific application depend on those PTMs?
💡 Rule of thumb: If your readout is a downstream signaling cascade in primary human cells, prioritize mammalian-expressed protein. If you are generating a calibration standard for mass spectrometry, E. coli may be perfectly adequate and significantly more cost-effective.
2. Purity Grade: Align Specification With Experimental Need
"High purity" is not a single number; it is a spectrum, and the right point on that spectrum depends entirely on what you are measuring and how sensitive your readout is.
- >95% purity (SDS-PAGE): Standard for most cell-based assays, ELISAs, and receptor binding studies.
- >98% purity: Required for SPR, ITC, crystallography, and rigorous kinetic measurements.
- GMP-grade: Mandatory for anything moving into clinical or preclinical in vivo models under regulatory oversight.
NOTE: Verification method matters too. SDS-PAGE gives a broad estimate; HPLC-SEC and mass spectrometry offer far higher resolution. When you purchase recombinant proteins for high-sensitivity work, always ask for the analytical method behind the purity number, as a "≥95% pure" claim backed by SDS-PAGE is not the same as one confirmed by HPLC.
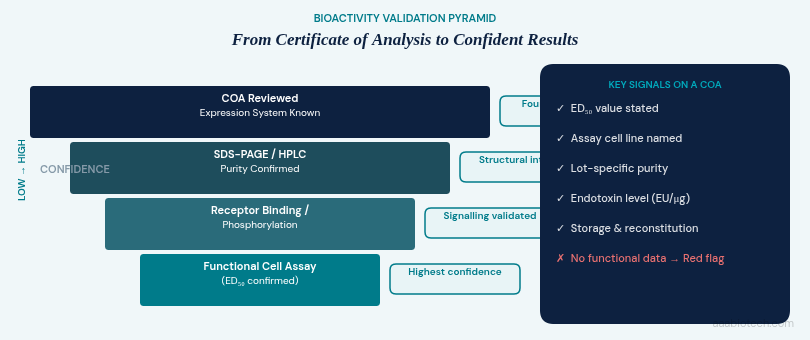
3. Bioactivity: The Metric That Predicts Real-World Performance
A protein can appear pure on a gel and still be biologically inert. Denaturation during purification or lyophilization, incorrect disulfide bridging, or aggregation can all produce a protein that looks clean on paper but fails in your assay. This is why functional validation data is non-negotiable.
For recombinant cytokines and growth factors, common bioactivity assays include:
- Cell proliferation/viability assays: e.g., measuring ED₅₀ for PDGF-BB on fibroblasts.
- Receptor binding/phosphorylation assays: e.g., STAT3 activation for IL-6.
- Chemotaxis assays: migration responses to CXCL chemokines.
- Enzymatic activity assays: for proteases and kinases.
When comparing vendors, do not stop at the purity specification. Pull the Certificate of Analysis (COA) and look for a stated ED₅₀ (effective dose for 50% maximal effect) alongside the assay format that generated it. If the COA offers no functional data, ask or look elsewhere.
4. Endotoxin Levels: The Variable That Quietly Ruins Experiments
Lipopolysaccharides (LPS) shed from the outer membrane of gram-negative bacteria, most notably E. coli, are the most common hidden contaminant in recombinant proteins. Even sub-nanogram quantities can trigger TLR4-mediated innate immune signaling in mammalian cell cultures, skewing cytokine readouts, confounding proliferation data, and invalidating in vivo results entirely.
Endotoxin thresholds vary by application:
- In vitro cell culture: < 1 EU/μg is the general benchmark.
- In vivo / preclinical: Often < 0.1 EU/μg; verified by Limulus Amebocyte Lysate (LAL) or recombinant Factor C (rFC) assay.
- GMP therapeutic: Strict regulatory limits per dose unit apply.
💡 If your target cells are immune lineage — macrophages, dendritic cells, T cells — then treat endotoxin control as a primary selection criterion, not a footnote.
5. Protein Tags: Useful Handles That Can Also Interfere
Affinity tags (His, GST, FLAG, Fc, Avi) are engineering tools that simplify purification and enable downstream detection. They are not biologically neutral, and their presence can affect receptor binding, dimerization, and intracellular signaling.
- His-tag: Small, minimally disruptive; good for most applications, but can interact with metal-chelating surfaces.
- GST-tag: Promotes solubility but is bulky, and often removed before functional assays.
- Fc-fusion: Extends half-life and aids detection; can cause artificial dimerization of the protein of interest.
- Tag-free: Optimal for in vivo studies and receptor-binding assays where steric interference is a concern.
When in doubt, consult published literature on the same protein; if successful studies used tag-free versions for your assay format, that is a strong signal as to what strategy should be aimed for.
6. Formulation, Storage Conditions, and Stability
A perfectly expressed and purified protein can lose activity before it ever reaches your cells if it is stored, handled, or reconstituted incorrectly. Buffer composition, carrier proteins, lyophilization quality, and freeze-thaw cycling all affect stability.
- Lyophilized vs liquid: Lyophilized formats offer longer shelf life and transport stability; liquids are ready to use but more susceptible to degradation.
- Carrier protein (BSA): Common in low-concentration formulations to prevent adsorption to tube walls, but BSA is not appropriate for serum-free or defined culture systems.
- Reconstitution: Always follow vendor instructions. Many cytokines and growth factors require reconstitution in sterile PBS + 0.1% BSA. Do not use plain water for lyophilized proteins.
- Aliquoting: Minimize freeze-thaw cycles. Single-use aliquots at −80 °C are the best practice for precious proteins.
7. Species Compatibility and Cross-Reactivity
Recombinant proteins are species-specific in most cases. Human IL-2 does not always bind mouse IL-2 receptor with the same affinity, and vice versa. Before finalizing your selection, confirm:
- The protein's species of origin matches your experimental model.
- If cross-reactivity is claimed, it is backed by binding or functional data, and not just homology.
- For xenograft or humanized mouse models, the correct species form is used for the target compartment of interest.
8. Supplier Reliability: Batch Consistency and Documentation
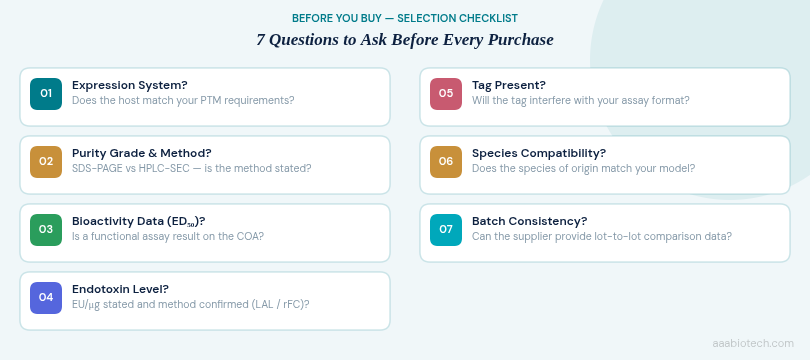
Science is reproducible only when reagents are reproducible. A supplier who cannot provide lot-to-lot consistency data, or whose COA documentation is sparse, is a reproducibility liability. When evaluating where to purchase recombinant proteins for ongoing programs, these are the non-negotiable supplier criteria:
- Lot-specific COA with purity, endotoxin, and bioactivity data.
- SDS-PAGE and/or HPLC trace included or available upon request.
- Clearly stated expiry, storage recommendations, and reconstitution protocol.
- Transparent manufacturing information (expression host, purification strategy, formulation).
- History of peer-reviewed citations for the product.
Putting It All Together
Rigorous protein selection is not bureaucracy; it is the foundation of reproducible, publishable science. By systematically evaluating expression systems, purity grade, bioactivity validation, endotoxin level, tag design, formulation, species compatibility, and supplier quality, researchers eliminate the most common sources of experimental failure before pipetting even begins. The extra ten minutes spent reading a COA carefully today saves days of troubleshooting tomorrow.
AAA Biotech's portfolio of research-grade proteins, including validated recombinant growth factors and cytokines across multiple expression platforms, is built around exactly these principles. Every product is manufactured with rigorous quality control, lot-specific documentation, and functional verification to support your most demanding applications.
- His-tag: Small, minimally disruptive; good for most applications, but can interact with metal-chelating surfaces.
Faq's
What is the best expression system for recombinant proteins used in cell culture?
For most cell-based assays, mammalian expression systems (HEK293 or CHO) are preferred because they produce proteins with human-like post-translational modifications, which are critical for accurate receptor binding and downstream signaling. However, well-validated E. coli-expressed proteins with confirmed bioactivity data can also perform reliably in many applications, and the COA should always be your guide.
How do I know if a recombinant protein is biologically active?
Look for an ED₅₀ value on the product's Certificate of Analysis (COA), tested in a cell-based functional assay relevant to the protein's biology; for example, a proliferation assay for growth factors or a phosphorylation assay for cytokines. If a vendor does not provide functional bioactivity data, this is a red flag.
What endotoxin level is acceptable for cell culture experiments?
For standard mammalian cell culture, an endotoxin level of less than 1 EU/μg is the broadly accepted benchmark. For sensitive immune cell work (macrophages, dendritic cells, T cells) or in vivo studies, levels below 0.1 EU/μg are recommended. Always verify this figure from the lot-specific COA, not just the general product specification.
Should I choose a tagged or tag-free recombinant protein?
It depends on your application. His-tagged proteins are convenient for pull-down assays and are minimally disruptive in many functional contexts. However, for receptor-binding studies, in vivo experiments, or applications where the tag's steric footprint could affect the protein's activity, tag-free formats are strongly preferred. Check whether published studies using the same protein used a tagged or tag-free version.
How should recombinant proteins be stored to maintain activity?
Most recombinant proteins should be stored at −80 °C in single-use aliquots to avoid activity loss from repeated freeze-thaw cycles. Lyophilized proteins must be reconstituted following the vendor's instructions, and typically in sterile PBS supplemented with a carrier protein such as 0.1% BSA — unless working in defined serum-free systems where BSA is incompatible. Avoid storing reconstituted protein at 4 °C for more than a few days.

 Cynthia
Cynthia