In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

5 Major Key Takeaways
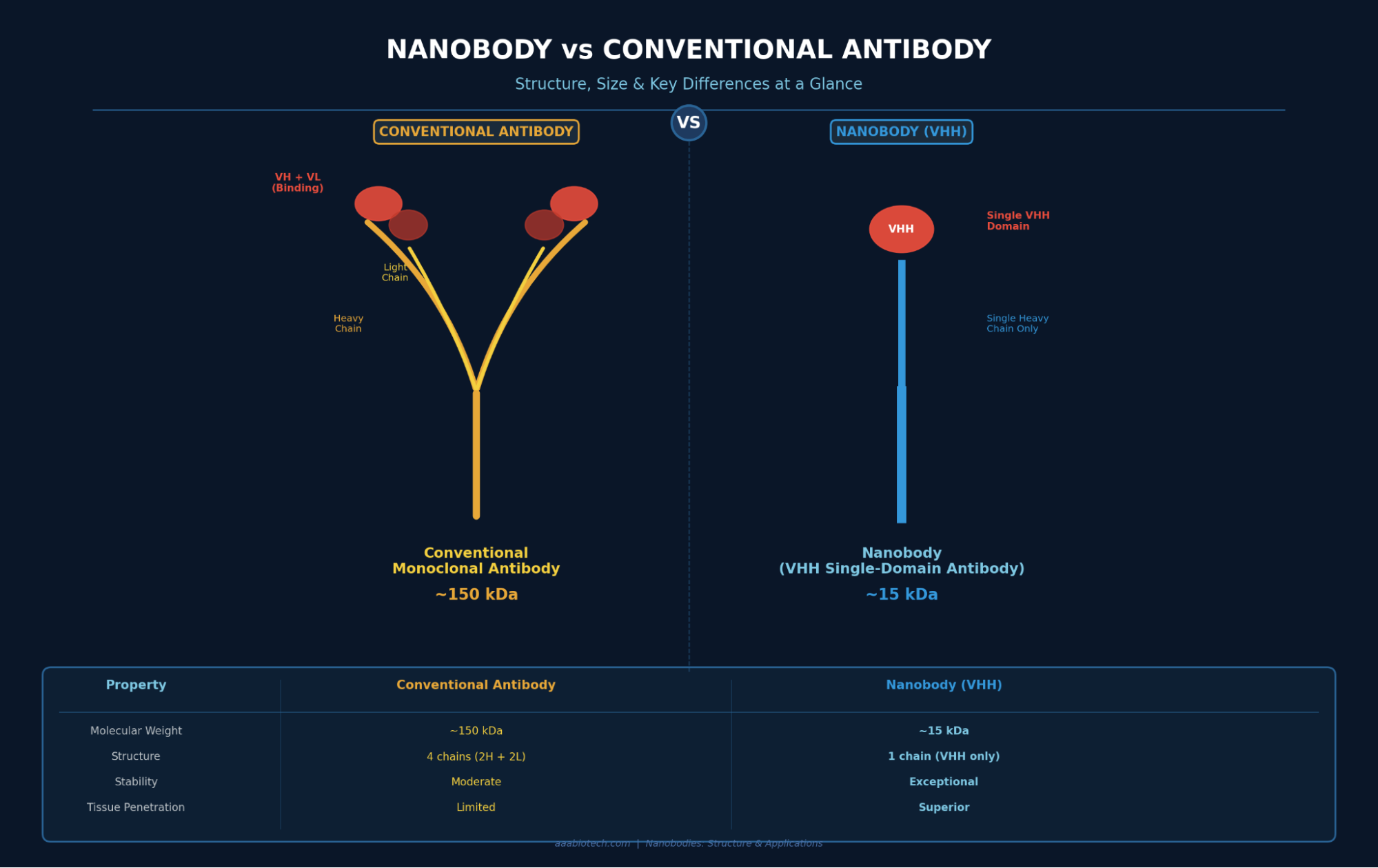
- Nanobodies are the smallest functional antibody fragments (~15 kDa), derived from camelid animals like llamas and camels.
- They consist of a single variable domain (VHH), unlike conventional antibodies that need two chains to bind an antigen.
- Their tiny size gives them superior tissue and tumor penetration compared to traditional monoclonal antibodies.
- Nanobodies are exceptionally stable under extreme temperature and pH conditions, which is a huge plus for diagnostics.
- From cancer therapy to super-resolution microscopy, their applications in research and medicine are rapidly expanding.
Picture a regular antibody, the classic Y-shaped protein your immune system deploys to fight off invaders. Now imagine taking just the very tip of that Y, stripping it down to one single compact unit, and having it work just as well, and actually, in many ways, even better. That's essentially what a nanobody is.
Discovered in the late 1980s by a group of researchers at the Vrije Universiteit Brussel (almost by accident, from a camel blood sample), nanobodies have quietly become one of the most exciting tools in modern biomedical science. Today, they're being used for everything from diagnosing infectious diseases to delivering cancer drugs directly to tumors.
So, what exactly are nanobodies, why do researchers reach for them instead of conventional antibodies, and where is this technology headed? Let's break it all down.
What Are Nanobodies, Exactly?
In immunology, a nanobody refers to the variable domain of a heavy-chain-only antibody — a unique type of antibody found exclusively in camelid animals (camels, llamas, dromedaries, and alpacas) and some sharks. These special antibodies have no light chains at all, which means their entire antigen-binding function is packed into a single small domain.
That single domain is called the VHH domain (Variable domain of the Heavy chain of Heavy-chain antibodies), and when it's isolated and used on its own, we call it a nanobody, also known as a single-domain antibody or VHH antibody.
Think of it this way: A conventional antibody is like a full Swiss Army knife with lots of parts working together. A nanobody is the single blade pulled out, i.e., compact, purposeful, and surprisingly capable.
Facts to Remember:
- Nanobody Size: ~15 kDa
- Full Antibody Size: ~150 kDa
- Smaller than mAbs: 10×
- Polypeptide Chain Only: 1
Nanobody Structure: Why the Simplicity Is the Point
The significance of nanobody structure goes beyond just being small. What makes VHH domains remarkable is how they compensate for the missing light chain.
In a conventional antibody, the antigen-binding site is formed by both a heavy chain and a light chain working together. When you remove the light chain, you'd expect the binding region to be compromised. But camelid VHH domains have evolved unique adaptations:
- Longer CDR3 loops are the part that directly contacts the antigen is extended, reaching into cavities and grooves that conventional antibodies physically cannot access.
- An extra disulfide bond adds structural stability without sacrificing flexibility.
- Hydrophilic substitutions at positions normally used to bind the light chain, thus preventing self-aggregation and making nanobodies water-soluble and easy to work with.
"Nanobodies can access hidden epitopes, including enzyme active sites, receptor clefts, and viral canyon regions that larger antibodies simply can't reach."
This combination makes single-domain antibodies not just smaller versions of normal antibodies; they're structurally unique tools with distinct targeting capabilities.
Nanobody vs Monoclonal Antibody: How They Stack Up
If you're deciding between a nanobody and a monoclonal antibody for your research, here's a quick, honest comparison:
| Properties | Monoclonal Antibody (mAb) | Nanobody (VHH) |
|---|---|---|
| Molecular Weight | ~150 kDa | ~15 kDa (Smaller) |
| Chain Structure | 2 heavy + 2 light chains | 1 VHH chain only (Simpler) |
| Thermal Stability | Moderate (denatures ~65°C) | High (stable up to ~80°C+) ; More stable |
| Tissue Penetration | Limited by size | Superior; crosses barriers easily (Better) |
| Production Host | Mammalian cells (complex) | Bacteria / Yeast (simpler, cheaper) |
| Antigen Access | Flat/surface epitopes | Cavities, grooves, hidden sites (Broader) |
| Half-life in blood | ~21 days | ~1–2 hours (can be extended) |
NOTE: The short half-life of nanobodies can actually be an advantage in imaging (faster clearance = cleaner images), though for therapeutic uses, it's something that needs to be engineered around.
The Core Advantages of Nanobodies in Research
When researchers ask about the benefits of using nanobodies, the list keeps growing. Here are the ones that matter most in day-to-day lab work:
S: Exceptional Stability
Nanobodies tolerate heat, harsh pH, and even detergents that would destroy conventional antibodies. They can be lyophilized and stored at room temperature.
P: Deep Tissue Penetration
Their tiny size allows them to diffuse into dense tissues, solid tumors, and even cross the blood-brain barrier, thus opening doors for neurological research.
E: Easy to Engineer
As a single gene product, nanobodies are straightforward to modify. Multi-specific constructs, fusion proteins, and drug conjugates are all simpler to build.
C: Cost-Effective Production
Nanobody production in bacterial or yeast systems is far cheaper and faster than the mammalian cell culture needed for most monoclonal antibodies.
H: Hidden Epitope Access
The elongated CDR3 loop lets nanobodies bind inside enzyme active sites and receptor grooves, which are sites completely inaccessible to conventional antibodies.
L: Low Immunogenicity
Humanized nanobodies show reduced immune reactions in therapeutic applications, making them promising drug candidates with better tolerability profiles.
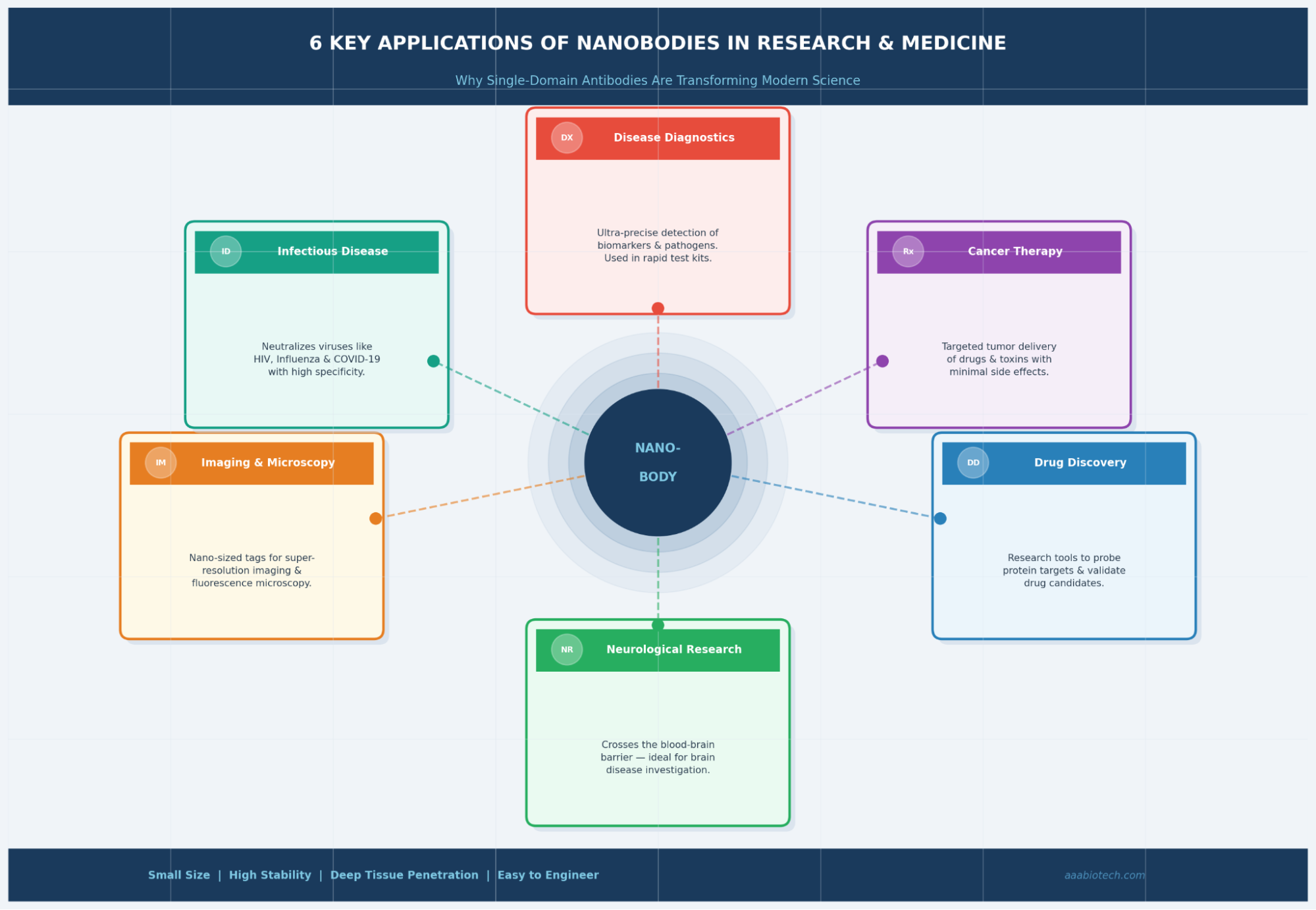
Where Are Nanobodies Being Used Today?
Nanobody applications in research and medicine have exploded over the past decade. Here's where scientists are putting them to work right now:
- Super-Resolution Microscopy (STORM, STED, PALM): Nanobodies are used as ultra-small labels that don't add much physical bulk to the target that critical for imaging structures at the nanometer scale. Products like anti-GFP or anti-RFP nanobodies have become standard microscopy tools.
- Cancer Theranostics: Loaded with drugs, toxins, or radioactive isotopes, nanobodies can seek out tumor cells and deliver payloads directly, far more precisely than systemic chemotherapy. Their fast clearance also reduces off-target radiation in PET imaging.
- Infectious Disease Diagnostics & Therapy: Nanobodies against SARS-CoV-2, HIV, RSV, and influenza have shown strong neutralization. They're also being explored in lateral flow and ELISA-based rapid diagnostic kits.
- Enzyme Inhibition & Protein Crystallography: Nanobodies act as conformational chaperones, locking proteins in specific states for structural studies. This has been invaluable in resolving GPCR crystal structures that were previously impossible.
- CAR-T Cell Engineering: VHH-based chimeric antigen receptors (CARs) are being developed as next-generation cell therapies, offering more compact and efficient tumor-targeting constructs.
- Neurological Research: Because nanobodies can cross the blood-brain barrier, they're being studied as both research tools and potential therapeutics for Alzheimer's disease, Parkinson's disease, and brain tumors.
How Are Nanobodies Made?
Nanobody production and purification are two of the reasons researchers love working with them. The general workflow proceeds as follows:
1. Immunization
A camelid animal (most commonly a llama or alpaca) is immunized with the target antigen over several weeks. The animal mounts an immune response that includes the production of VHH-containing heavy-chain antibodies.
2. Library Construction
RNA is extracted from peripheral blood lymphocytes, converted to cDNA, and the VHH sequences are amplified by PCR. These are cloned into a phage display vector to create a library of millions of nanobody candidates.
3. Phage Display & Selection
The phage library is panned against the target antigen through multiple rounds of selection, enriching for nanobodies that bind with high affinity and specificity.
4. Expression & Purification
Selected nanobody sequences are expressed in E. coli or yeast, and the protein is purified with the use of His-tag affinity chromatography to high purity in a matter of days. This ease of production is one of the clearest advantages of nanobodies over conventional antibodies, which require expensive mammalian cell systems.
Conclusion: AAA Biotech's Nanobody Research Tools
At AAA Biotech, we supply a growing catalog of validated nanobody-based reagents for research applications. Our product range includes:
Antibodies for pull-down assays, co-immunoprecipitation, and super-resolution microscopy, as well as target-specific VHH antibodies raised against key research proteins. Our nanobodies are recombinantly expressed, sequence-verified, and quality-controlled for binding performance.
If you're building a nanobody-drug conjugate, validating a new imaging protocol, or sourcing a reliable pull-down tool, our team is here to support your research with high-quality, well-characterized reagents.
Faq's
What exactly is a nanobody, and how is it different from a regular antibody?
A nanobody is the smallest functional antibody fragment — a single variable domain (VHH) derived from the heavy-chain-only antibodies found in camelid animals like llamas and camels. Unlike conventional antibodies that require two chains (heavy + light) to bind an antigen, a nanobody does the same job with just one compact chain, making it about 10 times smaller (~15 kDa vs ~150 kDa).
Why are nanobodies used in research instead of conventional antibodies?
Researchers choose nanobodies for several practical reasons: they're easier and cheaper to produce (in bacteria or yeast), remarkably stable under harsh conditions, small enough to penetrate dense tissues and reach hidden epitopes, and straightforward to engineer into fusion proteins or multi-specific formats. For imaging, diagnostics, and structural biology, especially, they outperform conventional antibodies in several key ways.
What are nanobodies used for in medicine?
Therapeutically, nanobodies are being explored for cancer treatment (targeted drug delivery), infectious disease (virus neutralization), neurological conditions (crossing the blood-brain barrier), and inflammation. Caplacizumab is a bivalently linked nanobody that became the first FDA-approved nanobody-based drug in 2019 for treating a rare blood-clotting disorder. Many more are in clinical trials.
Are nanobodies safe to use in humans?
Nanobodies can be humanized to reduce immune reactions, and the ones in clinical use so far have shown good tolerability. Because they're derived from camelid antibodies, they do have some sequence differences from human antibodies, but humanization strategies effectively address this. Their smaller size may also mean they clear faster from the body, which can be beneficial for imaging applications.
How are single-domain antibodies classified scientifically?
Single-domain antibodies are classified by the variable domain from which they originate. VHH antibodies come from the heavy chain of camelid antibodies; VNAR domains come from nurse sharks. Within these, nanobodies are further classified by their specificity (what antigen they target), their format (monovalent, bivalency, bispecific), and whether they are camelid-derived or fully humanized for therapeutic use.

 Cynthia
Cynthia