In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

5 Key Learnings from This Blog
- Recombinant antibody production uses cloned genetic sequences to eliminate reliance on animal hosts, offering reproducibility and scalability that traditional methods cannot match.
- Four major expression systems — bacterial, yeast, insect, and mammalian. Each presents unique trade-offs for yield, glycosylation, and complexity.
- High-throughput antibody production technologies like phage display and ribosome display enable rapid screening of vast antibody libraries.
- Rigorous quality control protocols, including SPR, ELISA, and SEC ensure binding specificity and structural integrity.
- Providers such as AAA Biotech play a critical role in bridging research-grade antibody engineering with GMP-compliant production pipelines.
The global antibody therapeutics market surpassed nearly $300 billion in 2025, and yet nearly 30% of research failures trace back to poorly characterized, batch-inconsistent antibodies. If a researcher is still relying on hybridoma-derived antibodies, then they are building on an unstable foundation.
Recombinant antibodies that are produced by cloning defined immunoglobulin gene sequences into expression systems eliminate that risk entirely.
They offer unlimited, sequence-verified supply, zero animal dependency, and engineering precision that conventional methods simply cannot match. Understanding how they're made isn't optional anymore. For competitive researchers and diagnostics teams, it's mission-critical.
Furthermore, these platforms support specialized formats like “bispecifics” and “nanobodies”, essential for advancing next-generation molecular diagnostics and precision therapeutics. Let us walk through it, step by step.
The Core Production Workflow
Recombinant antibody production begins with a well-defined antigen target. The antigen is typically a purified protein, peptide, or hapten presented in a format that drives a high-affinity immune response — or, in the case of fully in vitro systems, used directly to pan synthetic libraries.
Once candidate clones are identified through screening, the VH and VL gene sequences encoding the antigen-binding domains are cloned into expression vectors. These vectors are then introduced into a host expression system for protein production. The expressed antibody is subsequently purified and subjected to a battery of quality assays before release.
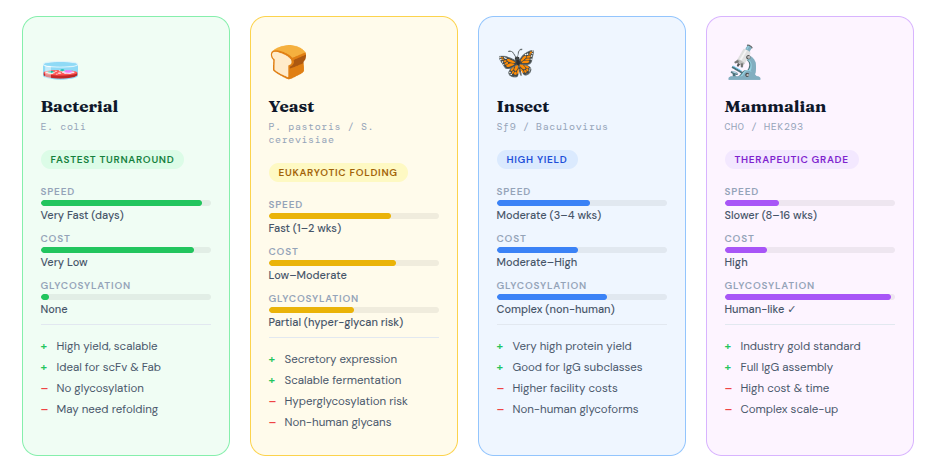
Choosing the Right Expression System
One of the most consequential decisions in antibody recombinant production is the choice of expression host. Each platform offers distinct advantages depending on the antibody format, required yield, and downstream application.
For most therapeutic-grade recombinant monoclonal antibody production, CHO (Chinese Hamster Ovary) cells remain the gold standard due to their capacity for proper glycosylation — a modification critical for Fc-mediated effector functions. For research reagents or rapid prototyping, bacterial or yeast systems offer faster, more cost-effective alternatives.
High-Throughput Antibody Production Technologies
Modern discovery pipelines depend on high-throughput antibody production methods to screen billions of clones in compressed timelines. The following platforms have become central to large-scale antibody discovery:
- Phage Display: Antibody gene libraries displayed on bacteriophage surfaces are panned against immobilized antigen. Multiple rounds enrich high-affinity binders, which are then sequenced and expressed independently.
- Yeast Display: Yeast surface display coupled with FACS enables quantitative affinity sorting, giving direct readout of binding strength and enabling fine-tuned affinity maturation.
- Ribosome Display: A fully in vitro platform that links mRNA, ribosome, and translated protein, enabling ultra-diverse library screening without transformation bottlenecks.
- Single B-Cell Sequencing: Captures native VH/VL pairing from antigen-specific B cells, preserving the immunological context of the original immune response.
AAA Biotech partners with labs to leverage phage display and single B-cell sequencing workflows to accelerate the transition from antigen hit to production-ready clone — without sacrificing specificity data or sequence documentation along the way.
Antibody Formats Enabled by Recombinant Technology
One of the underappreciated advantages of recombinant platforms is the ability to engineer novel antibody formats that would be impossible to produce through hybridoma methods alone:
- scFv (Single-chain variable fragment): VH and VL domains fused by a flexible linker — compact, ideal for intracellular and imaging applications.
- Fab fragments: Retain one antigen-binding arm, used in diagnostic kits and therapeutic contexts where reduced Fc-mediated signaling is preferred.
- Bispecific antibodies: Engineered to simultaneously bind two distinct epitopes, increasingly important in immuno-oncology.
- Nanobodies (VHH): Single-domain antibody fragments derived from camelid heavy-chain antibodies, notable for their small size and stability under harsh conditions.
- Full-length IgG: Complete immunoglobulin molecules with intact Fc region, produced primarily in mammalian systems for therapeutic applications.
Purification, Formulation & Quality Control
Downstream processing is as critical as expression. Even a perfectly expressed antibody can fail if purification introduces aggregates, endotoxin, or incorrect disulfide bonding. Standard antibody or recombinant protein production protocols employ:
- Affinity Chromatography (Protein A/G/L)
Primary capture step achieving >95% purity in a single pass for IgG formats - Size Exclusion Chromatography (SEC)
Separates monomeric antibody from aggregates and fragments; critical for therapeutic applications - Surface Plasmon Resonance (SPR)
Real-time kinetic binding analysis for KD, kon, and koff measurements - ELISA & Western Blot
Functional specificity testing under both native and denaturing conditions - Mass Spectrometry & Sequencing
Confirms amino acid sequence, post-translational modifications, and glycan composition
All of these steps together constitute a complete antibody production method that satisfies both research-grade and clinical-grade requirements. Our organization, AAA Bio, partners with labs to integrate these workflows into fully documented quality management systems, ensuring that sequence data, production records, and QC results accompany every lot.
Why Recombinant Antibodies Are the Future
The scientific community's shift toward recombinant platforms is not arbitrary. It reflects a clear set of technical and ethical advantages:
- Animal-free production: Fully synthetic libraries remove the need for immunization, aligning with 3Rs principles in research ethics.
- Unlimited supply: Unlike hybridoma cell lines, a defined DNA sequence can be re-expressed in any appropriate host at any time.
- Precise engineering: CDR grafting, Fc region modifications, and affinity maturation are all sequence-driven — allowing targeted optimization without random mutagenesis.
- Cross-species compatibility: Humanized or fully human antibodies minimize immunogenicity in therapeutic contexts.
- Regulatory traceability: Every lot is traceable to a defined sequence — a necessity for pharmaceutical-grade material and increasingly expected in research contexts too.
Conclusion
The move toward recombinant antibody production is not merely a preference but a necessity for the advancement of modern molecular diagnostics. By providing a sequence-defined, genetically stable, and highly customizable platform, recombinant technology ensures that the reagents used in critical assays are as precise as the science they support.
As automation and AI continue to mature, the barriers to accessing these premium reagents will continue to fall, paving the way for a more reproducible and innovative future in life sciences.
Faq's
What is the main difference between recombinant antibodies and monoclonal antibodies?
Monoclonal antibodies (mAbs) are typically produced using hybridoma technology, where antibody-secreting B cells are fused with immortal myeloma cells. Recombinant antibodies, by contrast, are produced by cloning the antibody genes into an expression vector and expressing them in a host cell. The key distinction is that recombinant antibodies do not require animal cell lines for ongoing production — just a stable DNA sequence, making them more reproducible, scalable, and amenable to engineering.
How long does recombinant antibody production take?
The timeline varies significantly depending on the platform and format. Using phage display against a well-characterized antigen, it is possible to identify candidate clones within 2–4 weeks. Full-scale production, purification, and QC validation in a mammalian expression system typically requires 8–16 weeks.
Which expression system is best for recombinant monoclonal antibody production?
There is no universally "best" system — the choice depends on the intended use. For full-length therapeutic IgGs requiring proper glycosylation, CHO or HEK293 mammalian cells are preferred. For research-grade scFvs or Fab fragments where glycosylation is less critical, E. coli or yeast systems provide faster and cheaper production. Insect cell systems offer a middle ground with high yields and complex glycan profiles.
Can recombinant antibodies be produced without using any animals?
Yes. Fully synthetic antibody libraries constructed from human immunoglobulin gene segments — such as those used in ribosome display or phage display against purified antigens — are entirely animal-free. These in vitro selection platforms are increasingly favored for their alignment with 3Rs ethical guidelines and their ability to generate antibodies against toxic or non-immunogenic antigens that are difficult to use in animal immunization.
What quality control tests are essential for recombinant antibody validation?
A comprehensive QC panel for recombinant antibodies typically includes: SDS-PAGE or capillary electrophoresis for purity and molecular weight confirmation, SEC-HPLC for aggregate analysis, ELISA and Western blot for functional specificity testing, surface plasmon resonance (SPR) for quantitative binding kinetics, endotoxin testing (LAL assay) for safety applications, and mass spectrometry for sequence and post-translational modification verification. The exact battery depends on the downstream use — research-grade lots require fewer tests than clinical or therapeutic-grade material.

 Cynthia
Cynthia