In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

Summary
- Phospho antibody western blots require specialized protocols that differ meaningfully from standard western blotting workflows — particularly around sample lysis and blocking.
- Recombinant phospho-specific antibodies offer superior lot-to-lot consistency, specificity, and sensitivity compared to conventional polyclonal antibodies.
- Phosphatase and protease inhibitors added during cell lysis are non-negotiable for preserving phosphorylation signals.
- TBS-based buffers must replace PBS in all incubation and washing steps to prevent phosphate ion interference with antibody binding.
- Proper controls — including phosphatase-treated lysates and total protein antibodies are essential for confirming phospho signal specificity and validity.
Detecting phosphorylated proteins by Western blot is one of the most rewarding and most frustrating techniques in cell biology research. Phosphorylation events govern virtually every major signaling cascade, from the MAPK and PI3K/AKT pathways to mTOR and NF-κB signaling. Capturing these modifications accurately is critical for understanding disease mechanisms, drug target validation, and biomarker discovery.
Yet for all its importance, phospho-protein western blotting is notoriously prone to weak signals, background noise, and false positives. The culprit is rarely poor technique alone — it's often the antibody itself, or more precisely, how antibody quality and the experimental protocol interact.
This guide walks through the essential steps to optimize your western blot workflow specifically for phospho antibodies, and explains why the shift toward recombinant antibody formats in recent years has fundamentally improved what researchers can achieve at the bench.
Why Phospho Western Blots Are Uniquely Challenging
Phosphorylation is a dynamic, transient post-translational modification. Unlike total protein detection, phospho western blotting must capture the protein in a specific, and often low-abundance, modified state. Several biological and technical factors conspire against clean detection:
- Phosphatase activity: Endogenous phosphatases are released the moment cells are lysed and will rapidly strip phosphate groups from your target unless actively inhibited.
- Low stoichiometry: Even in stimulated cells, only a fraction of any given protein population may be phosphorylated at a specific site at any moment.
- Epitope masking: SDS-PAGE denaturing conditions can alter the phospho-peptide conformation that the antibody recognizes.
- Buffer interference: Phosphate ions in PBS-based solutions compete with phospho-epitopes and can significantly reduce antibody binding efficiency.
- Antibody cross-reactivity: Traditional polyclonal antibodies raised against phospho-peptides frequently cross-react with the non-phosphorylated protein, confounding interpretation.
Addressing these challenges requires both a rigorous protocol and antibodies manufactured to the highest possible standard of specificity, which is where modern recombinant antibody production methods have made a tangible difference.
Step 1: Sample Preparation: Preserving the Phospho Signal
No antibody, however specific, can rescue a sample in which the phosphorylation has already been degraded. This makes lysis the single most critical step in a phospho western blot workflow.
Essential Lysis Buffer Components
- Phosphatase inhibitors: Include both serine/threonine and tyrosine phosphatase inhibitors (e.g., sodium fluoride, sodium orthovanadate, or a commercial cocktail) to prevent dephosphorylation.
- Protease inhibitors: Prevent target protein degradation, which also indirectly protects the phospho-epitope environment.
- Cold conditions: Keep all buffers and samples on ice throughout lysis. Pre-chill your centrifuge. Cold temperatures slow phosphatase and protease activity significantly.
- Speed: Move quickly from cell harvest to lysis buffer. The longer cells sit without inhibitors, the more phosphorylation is lost.
- Minimal lysis volume: Use the smallest practical volume of lysis buffer to concentrate your target protein, improving detection sensitivity.
"The phospho signal is perishable from the moment the cell membrane is disrupted. Optimal sample preparation is not just good practice — it is the experiment."
Step 2: Antibody Selection: Why Recombinant Matters
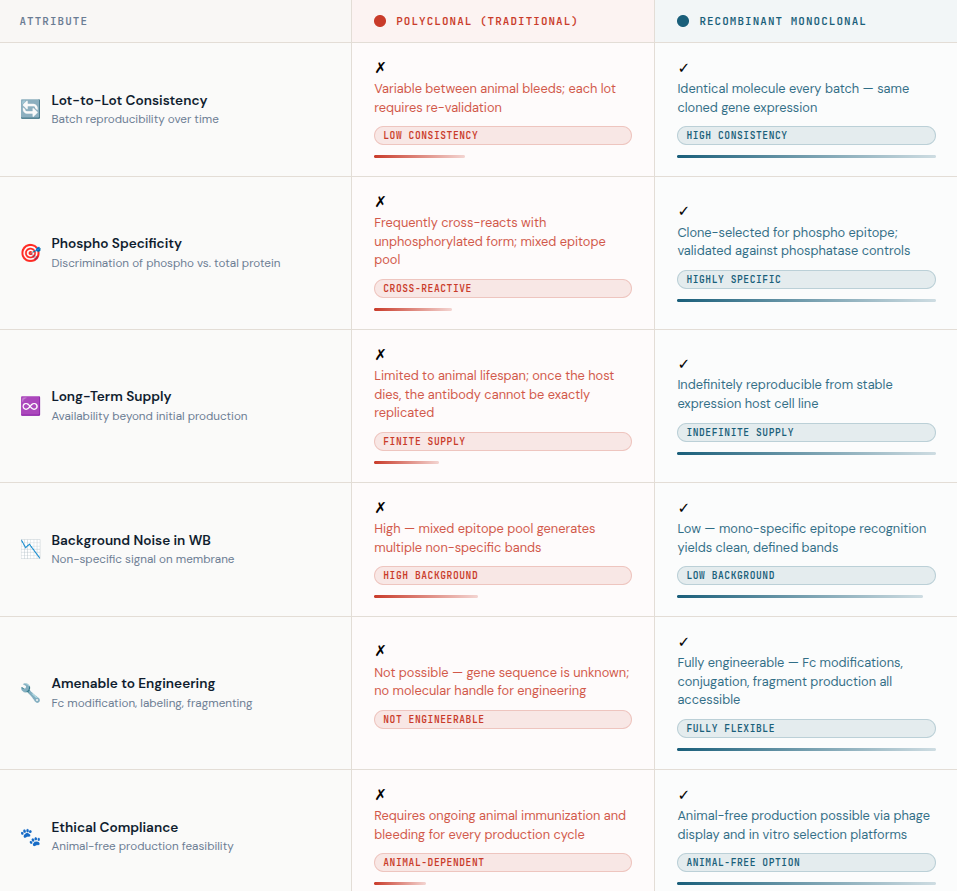
Antibody quality is the single largest variable in phospho Western blot performance. The traditional path of immunizing animals with phospho-peptides and collecting polyclonal serum has served the field for decades. But it carries fundamental limitations: lot-to-lot variability, declining specificity over time, and cross-reactivity with the unphosphorylated protein.
The shift toward recombinant antibodies addresses these limitations systematically. A recombinant antibody is produced in vitro from cloned, sequenced antibody genes expressed in mammalian cell lines — it is not harvested from an animal. This approach to antibody production offers several advantages that are particularly relevant for phospho detection:
- Defined sequence: Because the encoding genes are known and stored, the exact same antibody can be reproduced indefinitely with no genetic drift — a limitation that affects hybridoma-derived recombinant monoclonal antibody production workflows.
- Superior specificity: During the screening stages of recombinant antibody production, clones are rigorously selected for their ability to distinguish the phosphorylated form from the total protein.
- Scalability: High-throughput antibody production platforms — particularly CHO and HEK293 mammalian expression systems — allow manufacturing at scale without sacrificing quality.
- Animal-free options: Many antibody production methods for recombinant formats, including phage display and B-cell cloning, can proceed entirely in vitro, aligning with modern ethical research standards.
At AAA Biotech, the phospho-specific antibodies available for western blot applications are produced using validated recombinant monoclonal antibody production pipelines, ensuring the consistency and sensitivity that demanding phosphoprotein detection requires.
Step 3: Blocking and Buffer Choices
Buffer selection in phospho western blotting is not a matter of preference - it is a matter of chemistry. Two common protocol pitfalls undermine many experiments before the antibody even touches the membrane.
Avoid PBS — Use TBS-T Throughout
Standard phosphate-buffered saline (PBS) contains sodium phosphate, which directly competes with phospho-epitopes on your target protein for antibody binding. Replace PBS with Tris-buffered saline containing 0.1% Tween-20 (TBS-T) in all wash and incubation steps. This single change can dramatically improve signal-to-noise ratios.
Skip the Milk — Use BSA for Blocking
Nonfat dry milk is the default blocking reagent in many Western blot protocols. However, milk contains casein, a heavily phosphorylated protein that will compete with your phospho-target and reduce antibody signal significantly. Instead, use 5% BSA (bovine serum albumin) dissolved in TBS-T. Ensure the BSA is thoroughly dissolved — undissolved particles create speckles on the membrane.
Step 4: Controls That Cannot Be Skipped
Phospho western blot data without appropriate controls is difficult to interpret and nearly impossible to publish. The following controls should be considered standard practice:
- Stimulated vs. unstimulated lysates: Always include a positive control — cells treated with the relevant stimulus (growth factor, cytokine, kinase activator) alongside an unstimulated negative control. This confirms your antibody detects induced phosphorylation, not basal background.
- Phosphatase treatment control: Treat one aliquot of your lysate with lambda phosphatase or calf intestinal phosphatase before loading. If the antibody is phospho-specific, the band should disappear or dramatically reduce in the treated lane.
- Total protein antibody: Always pair every phospho-antibody detection with an antibody recognizing the total (phospho + non-phospho) form of the same protein. This allows you to normalize phosphorylation levels to protein expression and account for loading differences.
- Kinase inhibitor negative control: Where possible, pre-treat cells with a specific kinase inhibitor to block the relevant phosphorylation event. This provides a biologically validated negative control.
Step 5: Detection, Stripping, and Reprobing
When it comes to detection, sensitivity matters more in phospho Western blots than in most other antibody-based applications. Because phosphorylated protein is inherently less abundant than total protein, a highly sensitive chemiluminescent substrate — such as an enhanced ECL formulation — will often outperform standard HRP substrates in revealing faint phospho bands.
If your protocol requires detecting both the phospho-specific and total protein with antibodies of different molecular weights on the same blot, always probe for the phospho-protein first. Stripping protocols, even gentle ones, degrade the phospho-epitope over time and can make re-probing for phospho-protein after stripping unreliable.
For studies comparing multiple phosphorylation events simultaneously, fluorescent multiplexing is increasingly the preferred approach. It allows simultaneous detection of the phospho and total protein in different fluorescence channels on a single blot, eliminating stripping variability entirely and improving quantitative accuracy.
Conclusion: The Role of Recombinant Antibody Production in Research Reproducibility
The crisis of reproducibility in biomedical research has prompted widespread scrutiny of antibody quality. A key contributor to irreproducible western blot data is batch-to-batch variation in polyclonal antibody preparations — a problem that is structurally solved by switching to recombinant formats.
Understanding how recombinant antibodies are made helps researchers appreciate why the investment in higher-quality reagents pays dividends. The process — from antigen design through gene cloning, vector construction, mammalian expression, and rigorous validation is more controlled and traceable than traditional hybridoma or polyclonal routes.
Advances in high throughput antibody production platforms now mean that recombinant formats are available at competitive price points, making them accessible not only for therapeutic pipelines but for standard research applications. Antibody recombinant formats from suppliers committed to comprehensive validation — including western blot testing against phosphatase-treated and knockout controls — represent the current gold standard for phospho detection work.
Faq's
Why is my phospho antibody showing signals in both stimulated and unstimulated lanes?
This typically indicates one of three things: your antibody is cross-reacting with the non-phosphorylated form of the protein (a common limitation of polyclonal phospho antibodies), insufficient stimulation to clearly separate phosphorylation states, or insufficient washing steps allowing non-specific binding. To troubleshoot, run a phosphatase-treated lysate alongside your samples. If the signal persists after phosphatase treatment, the antibody is not phospho-specific. Switching to a validated recombinant monoclonal phospho antibody often resolves this issue by providing greater epitope selectivity.
Can I use milk as a blocking agent for phospho Western blots?
Generally, no. Milk contains casein, which is itself a heavily phosphorylated protein. When used as a blocking agent, casein competes with your phosphorylated target for antibody binding, resulting in dramatically reduced or absent phospho signals. The standard recommendation is to block with 5% BSA in TBS-T instead. Some phospho antibodies may be compatible with milk — always check the manufacturer's validated protocol, but BSA in TBS-T is the safest default.
Why do recombinant antibodies perform better for phospho detection?
For phospho detection, consistency of recombinant antibodies helps. The recombinant monoclonal phospho antibodies can be specifically selected for their ability to discriminate the phosphorylated from the non-phosphorylated epitope, delivering cleaner, more specific western blot results with less background.
How do I store lysates to protect phosphorylation signals?
For best results, store cell lysates at −80 °C rather than −20 °C. Lysates stored at −20 °C are more susceptible to freeze-thaw cycles and residual enzymatic activity that can degrade phosphorylation over time. Critically, always prepare lysates in the presence of fresh phosphatase and protease inhibitors, and transfer lysates into SDS sample buffer quickly if storing long-term. Avoid multiple freeze-thaw cycles, as each cycle risks progressive dephosphorylation even in inhibitor-containing buffers.
Should I probe for phospho-protein or total protein first when stripping and reprobing?
Always probe for the phospho-protein first. Stripping protocols — even mild, low-pH or reducing-agent-based strip buffers — progressively degrade the phospho-epitope on the membrane. If you probe for total protein first and then strip and re-probe for phospho-protein, there is a significant risk of false negatives or reduced signal intensity from the phospho antibody. Alternatively, consider fluorescent multiplexing, which allows simultaneous detection of both phospho and total protein in separate fluorescent channels on the same blot without any stripping.

 Cynthia
Cynthia