In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).
Key Takeaway
- Polyclonal antibodies bind to multiple sites on a single antigen, improving immune recognition.
- They aid researchers in identifying and validating the most effective vaccine targets early.
- Polyclonal antibodies are used extensively in both preclinical studies and clinical testing stages.
- They help measure comprehensive immune responses, providing accurate insights during vaccine trials.
- Polyclonal antibodies support long-term monitoring to ensure vaccines remain effective over time.
- They help manage pathogen mutations and variability, maintaining broad protection against changes.
- Polyclonal antibodies are widely applied in laboratory assays like ELISA and immunohistochemistry.
If you’ve ever wondered how antibodies and vaccine design actually come together, here’s the simple truth: it’s not just about creating a response, it’s about creating the right kind of response.
That’s where polyclonal antibodies come in.
Instead of targeting just one spot on a virus, they recognize multiple sites at once. This broader coverage is what makes vaccines more resilient. If one part of the pathogen mutates, the immune system still has other ways to recognize and fight it. That’s the foundation of a strong antibody vaccine strategy.
So, What Do Polyclonal Antibodies Actually Do?
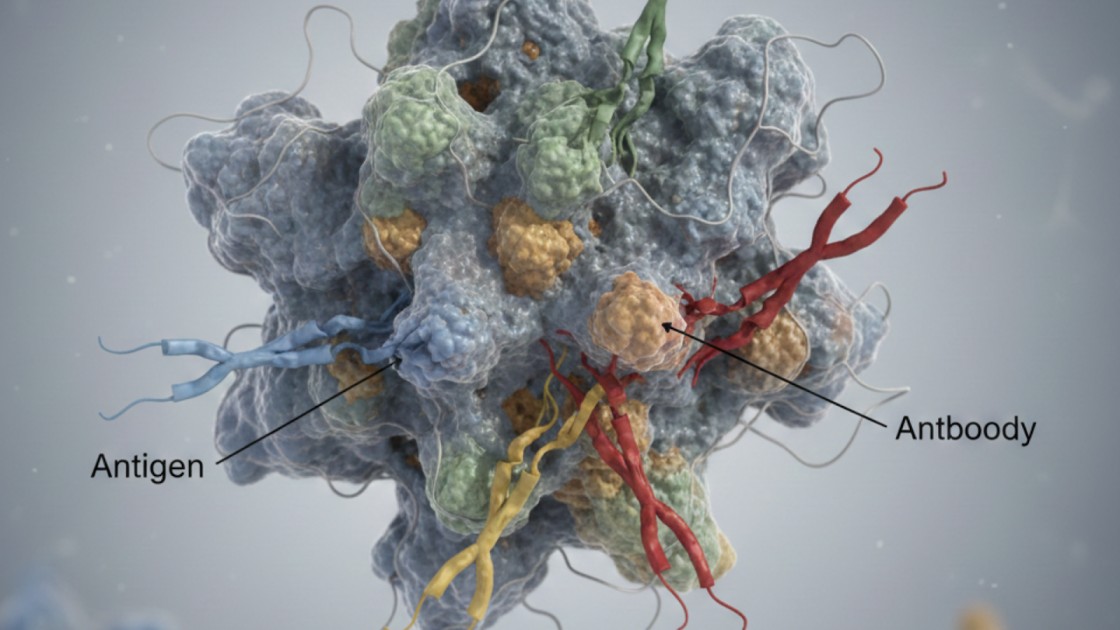
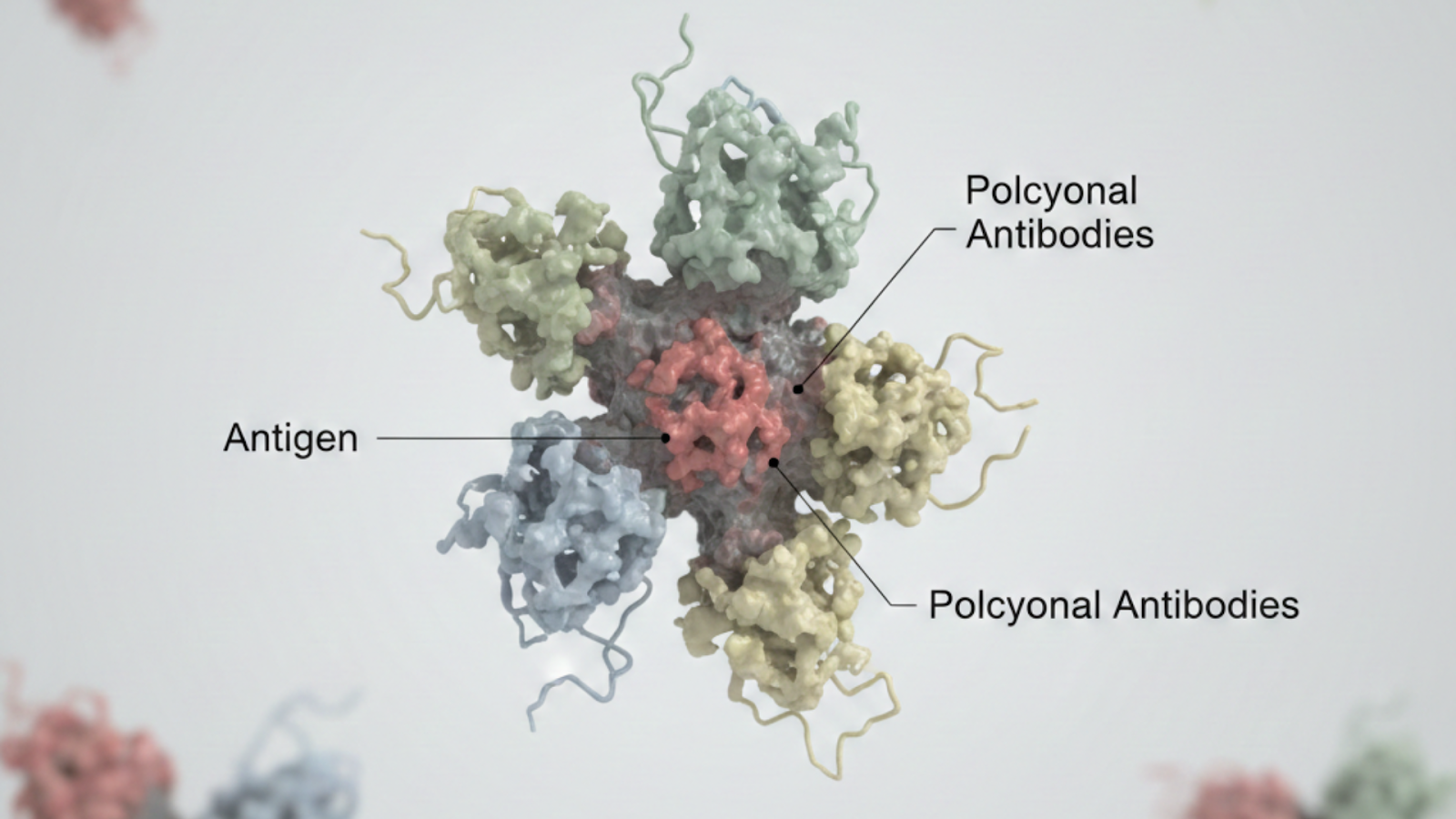
Polyclonal antibodies are a mixture of antibodies that recognize and bind to multiple sites (epitopes) on the same antigen.
These antibodies are involved throughout the vaccine journey, not just at one stage.
1. Antigen Discovery: They Help Identify The Right Target
Before anything else, researchers need to choose the right part of a pathogen to build a vaccine around. Polyclonal antibodies make this easier because they bind to multiple regions, helping confirm whether the target is actually visible and relevant to the immune system.
This is where the benefits of polyclonal antibodies in detecting target proteins really stand out. Even if the protein is present in low amounts or slightly altered, they’re still likely to detect it.
2. Preclinical Stage: They Validate Whether The Vaccine Idea Works
Once a target is selected, it needs to be tested. Polyclonal antibodies help confirm whether that target can trigger a meaningful immune response in animal models.
At this point, researchers aren’t looking for perfection—they’re looking for a realistic immune reaction. And that’s exactly what polyclonal antibodies reflect.
3. Clinical Stage: They Measure Real Immune Responses In Trials
When vaccines move into human trials, scientists need to know: is the vaccine actually working?
They don’t just look for one specific antibody. They measure the full polyclonal response, because that’s what real immunity looks like in the body—a mix of antibodies targeting different parts of the pathogen.
4. Post-Market Stage: They Support Long-Term Monitoring
Even after a vaccine is approved, the work doesn’t stop. Researchers continue tracking how people respond over time, especially as new variants appear.
Polyclonal antibodies help monitor whether the immune response stays broad and effective.
Did You Know?
Polyclonal antibodies are a growing business. The global market is expected to jump from $1.7 billion in 2024 to $2.8 billion by 2034. North America already owns nearly 40% of that market, and the numbers are only moving in one direction.
How Polyclonal Antibodies Help You Choose The Right Vaccine Target
What happens if the antigen that your vaccine is built around turns out to be structurally unstable or poorly accessible to the immune system?
You wind up finding out late in the process, after a lot of wasted (and likely expensive) work. This is exactly why antigen validation matters so much, and why polyclonal antibodies are the tool of choice at this stage.
Before a vaccine antigen is locked in, researchers need to answer three questions:
- Is the protein folding correctly?
- Are the right epitopes accessible on the surface?
- Could this antigen accidentally cross-react with something it was never meant to target?
Polyclonal antibodies help answer all of this in one go. Since they bind to multiple sites, they’re much better at picking up signals—even if the protein is present in low amounts or in a slightly altered form.
This is where the polyclonal antibodies really stand out in detecting target proteins. A single-target antibody might miss the protein if that one site is blocked. Polyclonal antibodies are far less likely to miss it.
Why This Matters Practically
If you get the target wrong here, it doesn’t just cost a few weeks. It can delay the entire project by years. That’s why polyclonal antibodies act like an early quality check before things move forward.
Quick Tip
If you’re testing multiple targets at once, using polyclonal antibodies in assays like ELISA or IHC can save a lot of time. You get faster, more reliable comparisons without running separate tests for each one.
How Polyclonal Antibodies Help You Measure Vaccine Efficacy
In trials, you’re not trying to confirm the presence of a single antibody. You’re trying to understand whether the vaccine is generating a “protective immune profile”.
That’s where the polyclonal response becomes critical.
A successful vaccine induces a diverse mix of antibodies targeting multiple epitopes. Measuring this response gives you a much more accurate view of efficacy compared to tracking a single, epitope-specific signal.
Why This Matters In Human Trials
At the clinical stage, the goal isn’t specificity; it’s coverage and durability.
Polyclonal antibodies give you a functional readout of whether the immune system is responding in a way that’s likely to hold up against variability and mutation.
In that sense, they’re not just a measurement tool; they define what “effective” immunity should look like in your trial.
A Real Example: Influenza Vaccination
In a controlled study, nine rabbits were immunized with type A influenza virions, and the epitope specificities of their antibody responses were analyzed. The polyclonal response was predominantly biased toward the site B epitope of the hemagglutinin, representing 65 to 82% of the total HI antibody in most animals. This pattern held regardless of rabbit strain, route of inoculation, or number of immunizing injections.
Why Batch Variability Still Matters In Polyclonal Antibody Work
If you’ve worked with pAbs, you already know batch variability isn’t just a small issue. It directly affects how reliable your data is.
What’s Driving The Variability?
At its core, it’s biological.
Since polyclonal antibodies come from living animals, every batch reflects a slightly different immune response. Even with tight protocols, you’ll still see variation because of:
- Differences between donor animals (age, health, immune history)
- Small changes in immunization setup (dose, schedule, adjuvants)
- Minor shifts during purification and processing
You can standardize the process, but you can’t fully standardize biology.
What This Means For Your Assays
In practice, a new batch can behave differently from the one you originally optimized with.
That becomes a real issue when you’re:
- Running high-throughput screening
- Comparing data across time points
- Maintaining consistency for regulatory or validation purposes
Instead of moving forward, you end up rechecking and readjusting—sometimes more than you’d like.
How Teams Are Managing Batch Variability
There’s no perfect fix yet, but a few approaches help reduce the impact of batch-to-batch differences:
- Pooling sera: balances out individual animal differences
- Standardized protocols: keep production more consistent
- Affinity purification: focuses on high-binding antibodies
- Recombinant polyclonal formats: an emerging option with better control
Final Thoughts
Polyclonal antibodies play a critical role across every stage of vaccine development—from identifying the right antigen to measuring real-world immune responses. Their ability to recognize multiple epitopes makes them especially valuable in building vaccines that remain effective despite mutations and variability. While challenges like batch variability exist, their overall contribution to research and validation remains indispensable.
For reliable polyclonal antibodies in vaccine research, AAA Bio offers high-quality, well-characterized solutions. Explore our range of products for accurate and efficient results.
Faq's
How long does it take to produce polyclonal antibodies?
Typically, four to eight weeks from immunization to usable serum, depending on the host animal and protocol. This is significantly faster than monoclonal antibody production, which can take several months, making pAbs a practical choice when timelines are tight.
Which animals are most commonly used to produce polyclonal antibodies?
Rabbits are the most widely used, accounting for nearly 43% of production globally. Goats, sheep, and chickens are also common. Each species offers different antibody yields, affinity levels, and practical advantages depending on the application.
Can polyclonal antibodies be used alongside monoclonal antibodies in the same study?
Yes, many research teams use both in parallel. Polyclonal antibodies are often used for broad initial screening, while monoclonals handle precise downstream targeting. Combining both gives researchers more complete and reliable data at different pipeline stages.
Are polyclonal antibodies used in cancer research, too?
Yes, though their role there is more limited compared to vaccine work. They are primarily used in cancer research for biomarker detection and tumor antigen characterization rather than as direct therapeutics, where monoclonal antibodies currently dominate.
What is the shelf life of polyclonal antibodies?
When stored correctly at minus 20 degrees Celsius or lower, polyclonal antibodies can remain stable for several years. Repeated freeze-thaw cycles are the most common cause of degradation, so aliquoting before storage is standard practice in most labs.

 Cynthia
Cynthia