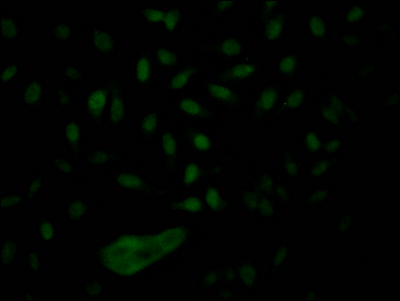
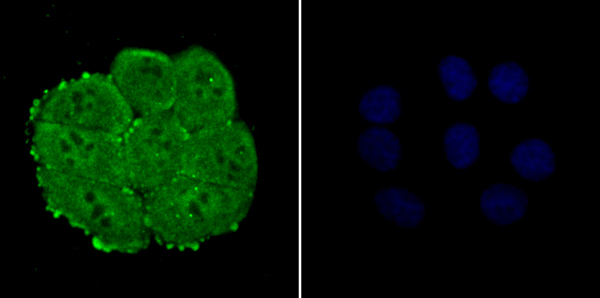
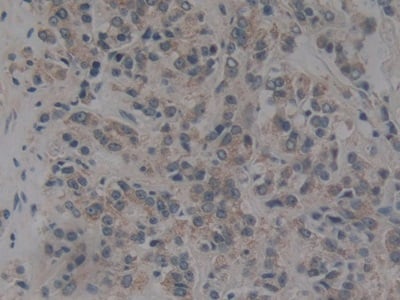
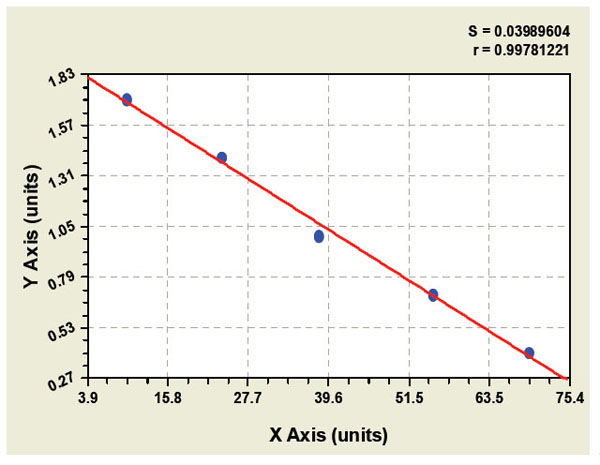
Nuclear pore complex protein Nup153 Recombinant Protein | NUP153 recombinant protein
Recombinant Human Nuclear pore complex protein Nup153
Gene Names
NUP153; N153; HNUP153
Purity
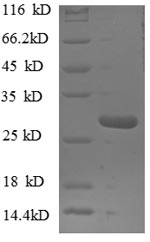
Greater or equal to 85% purity as determined by SDS-PAGE.
Synonyms
Nuclear pore complex protein Nup153; N/A; Recombinant Human Nuclear pore complex protein Nup153; 153 kDa nucleoporin; Nucleoporin Nup153; NUP153 recombinant protein
Host
E Coli or Yeast or Baculovirus or Mammalian Cell
Purity/Purification
Greater or equal to 85% purity as determined by SDS-PAGE.
Form/Format
Lyophilized or liquid (Format to be determined during the manufacturing process)
Sequence Positions
657-880aa; Partial
Sequence
KAGSSWQCDTCLLQNKVTDNKCIACQAAKLSPRDTAKQTGIETPNKSGKTTLSASGTGFGDKFKPVIGTWDCDTCLVQNKPEAIKCVACETPKPGTCVKRALTLTVVSESAETMTASSSSCTVTTGTLGFGDKFKRPIGSWECSVCCVSNNAEDNKCVSCMSEKPGSSVPASSSSTVPVSLPSGGSLGLEKFKKPEGSWDCELCLVQNKADSTKCLACESAKPG
Sequence Length
1,506
Preparation and Storage
Store at -20 degree C, for extended storage, conserve at -20 degree C or -80 degree C.
Related Product Information for NUP153 recombinant protein
Component of the nuclear pore complex (NPC), a complex required for the trafficking across the nuclear envelope. Functions as a scaffolding element in the nuclear phase of the NPC essential for normal nucleocytoplasmic transport of proteins and mRNAs. Involved in the quality control and retention of unspliced mRNAs in the nucleus; in association with TPR, regulates the nuclear export of unspliced mRNA species bearing constitutive transport element (CTE) in a NXF1- and KHDRBS1-independent manner. Mediates TPR anchoring to the nuclear membrane at NPC. The repeat-containing domain may be involved in anchoring other components of the NPC to the pore membrane. Possible DNA-binding subunit of the nuclear pore complex (NPC).
Product Categories/Family for NUP153 recombinant protein
References
Sequence analysis of a cDNA encoding a human nuclear pore complex protein, hnup153.McMorrow I., Bastos R., Horton H., Burke B.Biochim. Biophys. Acta 1217:219-223(1994) Complete sequencing and characterization of 21,243 full-length human cDNAs.Ota T., Suzuki Y., Nishikawa T., Otsuki T., Sugiyama T., Irie R., Wakamatsu A., Hayashi K., Sato H., Nagai K., Kimura K., Makita H., Sekine M., Obayashi M., Nishi T., Shibahara T., Tanaka T., Ishii S., Yamamoto J., Saito K., Kawai Y., Isono Y., Nakamura Y., Nagahari K., Murakami K., Yasuda T., Iwayanagi T., Wagatsuma M., Shiratori A., Sudo H., Hosoiri T., Kaku Y., Kodaira H., Kondo H., Sugawara M., Takahashi M., Kanda K., Yokoi T., Furuya T., Kikkawa E., Omura Y., Abe K., Kamihara K., Katsuta N., Sato K., Tanikawa M., Yamazaki M., Ninomiya K., Ishibashi T., Yamashita H., Murakawa K., Fujimori K., Tanai H., Kimata M., Watanabe M., Hiraoka S., Chiba Y., Ishida S., Ono Y., Takiguchi S., Watanabe S., Yosida M., Hotuta T., Kusano J., Kanehori K., Takahashi-Fujii A., Hara H., Tanase T.-O., Nomura Y., Togiya S., Komai F., Hara R., Takeuchi K., Arita M., Imose N., Musashino K., Yuuki H., Oshima A., Sasaki N., Aotsuka S., Yoshikawa Y., Matsunawa H., Ichihara T., Shiohata N., Sano S., Moriya S., Momiyama H., Satoh N., Takami S., Terashima Y., Suzuki O., Nakagawa S., Senoh A., Mizoguchi H., Goto Y., Shimizu F., Wakebe H., Hishigaki H., Watanabe T., Sugiyama A., Takemoto M., Kawakami B., Yamazaki M., Watanabe K., Kumagai A., Itakura S., Fukuzumi Y., Fujimori Y., Komiyama M., Tashiro H., Tanigami A., Fujiwara T., Ono T., Yamada K., Fujii Y., Ozaki K., Hirao M., Ohmori Y., Kawabata A., Hikiji T., Kobatake N., Inagaki H., Ikema Y., Okamoto S., Okitani R., Kawakami T., Noguchi S., Itoh T., Shigeta K., Senba T., Matsumura K., Nakajima Y., Mizuno T., Morinaga M., Sasaki M., Togashi T., Oyama M., Hata H., Watanabe M., Komatsu T., Mizushima-Sugano J., Satoh T., Shirai Y., Takahashi Y., Nakagawa K., Okumura K., Nagase T., Nomura N., Kikuchi H., Masuho Y., Yamashita R., Nakai K., Yada T., Nakamura Y., Ohara O., Isogai T., Sugano S.Nat. Genet. 36:40-45(2004) Preparation of a set of expression-ready clones of mammalian long cDNAs encoding large proteins by the ORF trap cloning method.Nakajima D., Saito K., Yamakawa H., Kikuno R.F., Nakayama M., Ohara R., Okazaki N., Koga H., Nagase T., Ohara O. The DNA sequence and analysis of human chromosome 6.Mungall A.J., Palmer S.A., Sims S.K., Edwards C.A., Ashurst J.L., Wilming L., Jones M.C., Horton R., Hunt S.E., Scott C.E., Gilbert J.G.R., Clamp M.E., Bethel G., Milne S., Ainscough R., Almeida J.P., Ambrose K.D., Andrews T.D., Ashwell R.I.S., Babbage A.K., Bagguley C.L., Bailey J., Banerjee R., Barker D.J., Barlow K.F., Bates K., Beare D.M., Beasley H., Beasley O., Bird C.P., Blakey S.E., Bray-Allen S., Brook J., Brown A.J., Brown J.Y., Burford D.C., Burrill W., Burton J., Carder C., Carter N.P., Chapman J.C., Clark S.Y., Clark G., Clee C.M., Clegg S., Cobley V., Collier R.E., Collins J.E., Colman L.K., Corby N.R., Coville G.J., Culley K.M., Dhami P., Davies J., Dunn M., Earthrowl M.E., Ellington A.E., Evans K.A., Faulkner L., Francis M.D., Frankish A., Frankland J., French L., Garner P., Garnett J., Ghori M.J., Gilby L.M., Gillson C.J., Glithero R.J., Grafham D.V., Grant M., Gribble S., Griffiths C., Griffiths M.N.D., Hall R., Halls K.S., Hammond S., Harley J.L., Hart E.A., Heath P.D., Heathcott R., Holmes S.J., Howden P.J., Howe K.L., Howell G.R., Huckle E., Humphray S.J., Humphries M.D., Hunt A.R., Johnson C.M., Joy A.A., Kay M., Keenan S.J., Kimberley A.M., King A., Laird G.K., Langford C., Lawlor S., Leongamornlert D.A., Leversha M., Lloyd C.R., Lloyd D.M., Loveland J.E., Lovell J., Martin S., Mashreghi-Mohammadi M., Maslen G.L., Matthews L., McCann O.T., McLaren S.J., McLay K., McMurray A., Moore M.J.F., Mullikin J.C., Niblett D., Nickerson T., Novik K.L., Oliver K., Overton-Larty E.K., Parker A., Patel R., Pearce A.V., Peck A.I., Phillimore B.J.C.T., Phillips S., Plumb R.W., Porter K.M., Ramsey Y., Ranby S.A., Rice C.M., Ross M.T., Searle S.M., Sehra H.K., Sheridan E., Skuce C.D., Smith S., Smith M., Spraggon L., Squares S.L., Steward C.A., Sycamore N., Tamlyn-Hall G., Tester J., Theaker A.J., Thomas D.W., Thorpe A., Tracey A., Tromans A., Tubby B., Wall M., Wallis J.M., West A.P., White S.S., Whitehead S.L., Whittaker H., Wild A., Willey D.J., Wilmer T.E., Wood J.M., Wray P.W., Wyatt J.C., Young L., Younger R.M., Bentley D.R., Coulson A., Durbin R.M., Hubbard T., Sulston J.E., Dunham I., Rogers J., Beck S.Nature 425:805-811(2003) Bienvenut W.V., Dozynkiewicz M., Norman J.C.Submitted (MAR-2009) to UniProtKB Exportin-5, a novel karyopherin, mediates nuclear export of double-stranded RNA binding proteins.Brownawell A.M., Macara I.G.J. Cell Biol. 156:53-64(2002) Tpr is localized within the nuclear basket of the pore complex and has a role in nuclear protein export.Frosst P., Guan T., Subauste C., Hahn K., Gerace L.J. Cell Biol. 156:617-630(2002) Direct interaction with nup153 mediates binding of Tpr to the periphery of the nuclear pore complex.Hase M.E., Cordes V.C.Mol. Biol. Cell 14:1923-1940(2003) Nucleoporins as components of the nuclear pore complex core structure and Tpr as the architectural element of the nuclear basket.Krull S., Thyberg J., Bjorkroth B., Rackwitz H.R., Cordes V.C.Mol. Biol. Cell 15:4261-4277(2004) Global, in vivo, and site-specific phosphorylation dynamics in signaling networks.Olsen J.V., Blagoev B., Gnad F., Macek B., Kumar C., Mortensen P., Mann M.Cell 127:635-648(2006) A probability-based approach for high-throughput protein phosphorylation analysis and site localization.Beausoleil S.A., Villen J., Gerber S.A., Rush J., Gygi S.P.Nat. Biotechnol. 24:1285-1292(2006) Combining protein-based IMAC, peptide-based IMAC, and MudPIT for efficient phosphoproteomic analysis.Cantin G.T., Yi W., Lu B., Park S.K., Xu T., Lee J.-D., Yates J.R. IIIJ. Proteome Res. 7:1346-1351(2008) Kinase-selective enrichment enables quantitative phosphoproteomics of the kinome across the cell cycle.Daub H., Olsen J.V., Bairlein M., Gnad F., Oppermann F.S., Korner R., Greff Z., Keri G., Stemmann O., Mann M.Mol. Cell 31:438-448(2008) A quantitative atlas of mitotic phosphorylation.Dephoure N., Zhou C., Villen J., Beausoleil S.A., Bakalarski C.E., Elledge S.J., Gygi S.P.Proc. Natl. Acad. Sci. U.S.A. 105:10762-10767(2008) Lys-N and trypsin cover complementary parts of the phosphoproteome in a refined SCX-based approach.Gauci S., Helbig A.O., Slijper M., Krijgsveld J., Heck A.J., Mohammed S.Anal. Chem. 81:4493-4501(2009) Integrase interacts with nucleoporin NUP153 to mediate the nuclear import of human immunodeficiency virus type 1.Woodward C.L., Prakobwanakit S., Mosessian S., Chow S.A.J. Virol. 83:6522-6533(2009) Quantitative phosphoproteomic analysis of T cell receptor signaling reveals system-wide modulation of protein-protein interactions.Mayya V., Lundgren D.H., Hwang S.-I., Rezaul K., Wu L., Eng J.K., Rodionov V., Han D.K.Sci. Signal. 2:RA46-RA46(2009) Lysine acetylation targets protein complexes and co-regulates major cellular functions.Choudhary C., Kumar C., Gnad F., Nielsen M.L., Rehman M., Walther T.C., Olsen J.V., Mann M.Science 325:834-840(2009) Protein Tpr is required for establishing nuclear pore-associated zones of heterochromatin exclusion.Krull S., Dorries J., Boysen B., Reidenbach S., Magnius L., Norder H., Thyberg J., Cordes V.C.EMBO J. 29:1659-1673(2010) Nucleoporin translocated promoter region (Tpr) associates with dynein complex, preventing chromosome lagging formation during mitosis.Nakano H., Funasaka T., Hashizume C., Wong R.W.J. Biol. Chem. 285:10841-10849(2010) Nucleoporin 153 arrests the nuclear import of hepatitis B virus capsids in the nuclear basket.Schmitz A., Schwarz A., Foss M., Zhou L., Rabe B., Hoellenriegel J., Stoeber M., Pante N., Kann M.PLoS Pathog. 6:E1000741-E1000741(2010) Extensive crosstalk between O-GlcNAcylation and phosphorylation regulates cytokinesis.Wang Z., Udeshi N.D., Slawson C., Compton P.D., Sakabe K., Cheung W.D., Shabanowitz J., Hunt D.F., Hart G.W.Sci. Signal. 3:RA2-RA2(2010) Quantitative phosphoproteomics reveals widespread full phosphorylation site occupancy during mitosis.Olsen J.V., Vermeulen M., Santamaria A., Kumar C., Miller M.L., Jensen L.J., Gnad F., Cox J., Jensen T.S., Nigg E.A., Brunak S., Mann M.Sci. Signal. 3:RA3-RA3(2010) Initial characterization of the human central proteome.Burkard T.R., Planyavsky M., Kaupe I., Breitwieser F.P., Buerckstuemmer T., Bennett K.L., Superti-Furga G., Colinge J.BMC Syst. Biol. 5:17-17(2011) System-wide temporal characterization of the proteome and phosphoproteome of human embryonic stem cell differentiation.Rigbolt K.T., Prokhorova T.A., Akimov V., Henningsen J., Johansen P.T., Kratchmarova I., Kassem M., Mann M., Olsen J.V., Blagoev B.Sci. Signal. 4:RS3-RS3(2011) Hikeshi, a nuclear import carrier for hsp70s, protects cells from heat shock-induced nuclear damage.Kose S., Furuta M., Imamoto N.Cell 149:578-589(2012) Localization of nucleoporin Tpr to the nuclear pore complex is essential for Tpr mediated regulation of the export of unspliced RNA.Rajanala K., Nandicoori V.K.PLoS ONE 7:E29921-E29921(2012) N-terminal acetylome analyses and functional insights of the N-terminal acetyltransferase NatB.Van Damme P., Lasa M., Polevoda B., Gazquez C., Elosegui-Artola A., Kim D.S., De Juan-Pardo E., Demeyer K., Hole K., Larrea E., Timmerman E., Prieto J., Arnesen T., Sherman F., Gevaert K., Aldabe R.Proc. Natl. Acad. Sci. U.S.A. 109:12449-12454(2012) An enzyme assisted RP-RPLC approach for in-depth analysis of human liver phosphoproteome.Bian Y., Song C., Cheng K., Dong M., Wang F., Huang J., Sun D., Wang L., Ye M., Zou H.J. Proteomics 96:253-262(2014) Solution structure of the second and third ZF-RANBP domains from human nuclear pore complex protein NUP153.RIKEN structural genomics initiative (RSGI) Submitted (AUG-2007) to the PDB data bankSolution structure of the second ZF-ranbp domain from human nuclear pore complex protein nup153.RIKEN structural genomics initiative (RSGI) Submitted (FEB-2009) to the PDB data bank+Additional computationally mapped references.<p>Provides general information on the entry.
NCBI and Uniprot Product Information
NCBI GI #
NCBI GeneID
NCBI Accession #
NCBI GenBank Nucleotide #
Molecular Weight
27.3 kDa
NCBI Official Full Name
nuclear pore complex protein Nup153 isoform 1
NCBI Official Synonym Full Names
nucleoporin 153kDa
NCBI Official Symbol
NUP153
NCBI Official Synonym Symbols
N153; HNUP153
NCBI Protein Information
nuclear pore complex protein Nup153
UniProt Protein Name
Nuclear pore complex protein Nup153
UniProt Gene Name
NUP153
UniProt Entry Name
NU153_HUMAN
Customer Reviews
Loading reviews...
Share Your Experience
Similar Products
Product Notes
The NUP153 nup153 (Catalog #AAA113207) is a Recombinant Protein produced from E Coli or Yeast or Baculovirus or Mammalian Cell and is intended for research purposes only. The product is available for immediate purchase. The immunogen sequence is 657-880aa; Partial. The amino acid sequence is listed below: KAGSSWQCDT CLLQNKVTDN KCIACQAAKL SPRDTAKQTG IETPNKSGKT TLSASGTGFG DKFKPVIGTW DCDTCLVQNK PEAIKCVACE TPKPGTCVKR ALTLTVVSES AETMTASSSS CTVTTGTLGF GDKFKRPIGS WECSVCCVSN NAEDNKCVSC MSEKPGSSVP ASSSSTVPVS LPSGGSLGLE KFKKPEGSWD CELCLVQNKA DSTKCLACES AKPG. It is sometimes possible for the material contained within the vial of "Nuclear pore complex protein Nup153, Recombinant Protein" to become dispersed throughout the inside of the vial, particularly around the seal of said vial, during shipment and storage. We always suggest centrifuging these vials to consolidate all of the liquid away from the lid and to the bottom of the vial prior to opening. Please be advised that certain products may require dry ice for shipping and that, if this is the case, an additional dry ice fee may also be required.Precautions
All products in the AAA Biotech catalog are strictly for research-use only, and are absolutely not suitable for use in any sort of medical, therapeutic, prophylactic, in-vivo, or diagnostic capacity. By purchasing a product from AAA Biotech, you are explicitly certifying that said products will be properly tested and used in line with industry standard. AAA Biotech and its authorized distribution partners reserve the right to refuse to fulfill any order if we have any indication that a purchaser may be intending to use a product outside of our accepted criteria.Disclaimer
Though we do strive to guarantee the information represented in this datasheet, AAA Biotech cannot be held responsible for any oversights or imprecisions. AAA Biotech reserves the right to adjust any aspect of this datasheet at any time and without notice. It is the responsibility of the customer to inform AAA Biotech of any product performance issues observed or experienced within 30 days of receipt of said product. To see additional details on this or any of our other policies, please see our Terms & Conditions page.Item has been added to Shopping Cart
If you are ready to order, navigate to Shopping Cart and get ready to checkout.