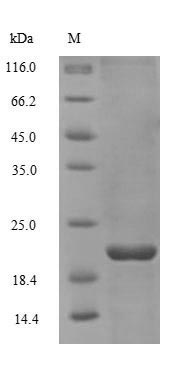
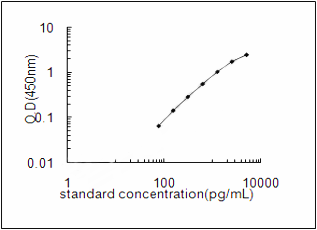
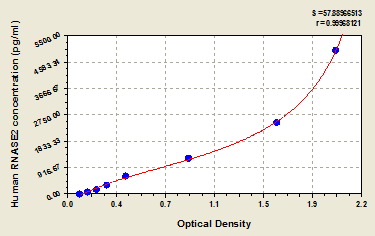
Non-secretory ribonuclease Recombinant Protein | RNASE2 recombinant protein
Recombinant Human Non-secretory ribonuclease
Gene Names
RNASE2; EDN; RAF3; RNS2
Purity
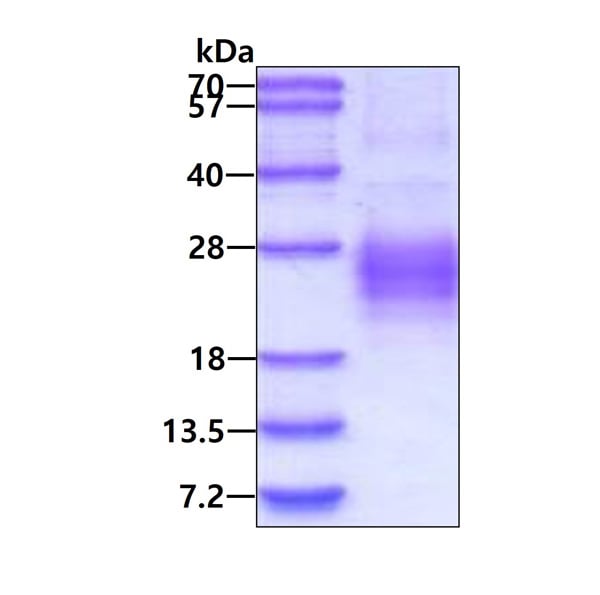
Greater or equal to 85% purity as determined by SDS-PAGE.
Synonyms
Non-secretory ribonuclease; N/A; Recombinant Human Non-secretory ribonuclease; Eosinophil-derived neurotoxin; RNase UpI-2; Ribonuclease 2; RNase 2; Ribonuclease US; RNASE2 recombinant protein
Host
E Coli or Yeast or Baculovirus or Mammalian Cell
Purity/Purification
Greater or equal to 85% purity as determined by SDS-PAGE.
Form/Format
Lyophilized or liquid (Format to be determined during the manufacturing process)
Sequence Positions
28-161aa; Full Length of Mature Protein
Sequence
KPPQFTWAQWFETQHINMTSQQCTNAMQVINNYQRRCKNQNTFLLTTFANVVNVCGNPNMTCPSNKTRKNCHHSGSQVPLIHCNLTTPSPQNISNCRYAQTPANMFYIVACDNRDQRRDPPQYPVVPVHLDRII
Preparation and Storage
Store at -20 degree C, for extended storage, conserve at -20 degree C or -80 degree C.
Related Product Information for RNASE2 recombinant protein
This is a non-secretory ribonuclease. It is a pyrimidine specific nuclease with a slight preference for U. Cytotoxin and helminthotoxin. Selectively chotactic for dendritic cells. Possesses a wide variety of biological activities.
Product Categories/Family for RNASE2 recombinant protein
References
Sequence of human eosinophil-derived neurotoxin cDNA identity of deduced amino acid sequence with human nonsecretory ribonucleases.Hamann K.J., Barker R.L., Loegering D.A., Pease L.R., Gleich G.J.Gene 83:161-167(1989) Eosinophil cationic protein cDNA. Comparison with other toxic cationic proteins and ribonucleases.Barker R.L., Loegering D.A., Ten R.M., Hamann K.J., Pease L.R., Gleich G.J.J. Immunol. 143:952-955(1989) Molecular cloning of the human eosinophil-derived neurotoxin a member of the ribonuclease gene family.Rosenberg H.F., Tenen D.G., Ackerman S.J.Proc. Natl. Acad. Sci. U.S.A. 86:4460-4464(1989) Structure and chromosome localization of the human eosinophil-derived neurotoxin and eosinophil cationic protein genes evidence for intronless coding sequences in the ribonuclease gene superfamily.Hamann K.J., Ten R.M., Loegering D.A., Jenkins R.B., Heise M.T., Schad C.R., Pease L.R., Gleich G.J., Barker R.L.Genomics 7:535-546(1990) Sequence variation at two eosinophil-associated ribonuclease loci in humans.Zhang J., Rosenberg H.F.Genetics 156:1949-1958(2000) Simonsen C.C., Kennedy J., Comstock L., Ashton N., McGrogan M. Amino acid sequence of the nonsecretory ribonuclease of human urine.Beintema J.J., Hofsteenge J., Iwama M., Morita T., Ohgi K., Irie M., Sugiyama R.H., Schieven G.L., Dekker C.A., Glitz D.G.Biochemistry 27:4530-4538(1988) Biochemical and functional similarities between human eosinophil-derived neurotoxin and eosinophil cationic protein homology with ribonuclease.Gleich G.J., Loegering D.A., Bell M.P., Checkel J.L., Ackerman S.J., McKean D.J.Proc. Natl. Acad. Sci. U.S.A. 83:3146-3150(1986) Purification and properties of bovine kidney ribonucleases.Niwata Y., Ohgi K., Sanda A., Takizawa Y., Irie M.J. Biochem. 97:923-934(1985) Purification and characterization of a ribonuclease from human liver.Sorrentino S., Tucker G.K., Glitz D.G.J. Biol. Chem. 263:16125-16131(1988) Characterization of a unique nonsecretory ribonuclease from urine of pregnant women.Sakakibara R., Hashida K., Kitahara T., Ishiguro M.J. Biochem. 111:325-330(1992) Characterisation of UGP and its relationship with beta-core fragment.Kardana A., Bagshawe K.D., Coles B., Read D., Taylor M.Br. J. Cancer 67:686-692(1993) New type of linkage between a carbohydrate and a protein C-glycosylation of a specific tryptophan residue in human RNase Us.Hofsteenge J., Mueller D.R., de Beer T., Loeffler A., Richter W.J., Vliegenthart J.F.G.Biochemistry 33:13524-13530(1994) The hexopyranosyl residue that is C-glycosidically linked to the side chain of tryptophan-7 in human RNase Us is alpha-mannopyranose.de Beer T., Vliegenthart J.F.G., Loeffler A., Hofsteenge J.Biochemistry 34:11785-11789(1995) Recognition signal for C-mannosylation of Trp-7 in RNase 2 consists of sequence Trp-x-x-Trp.Krieg J., Hartmann S., Vicentini A., Glasner W., Hess D., Hofsteenge J.Mol. Biol. Cell 9:301-309(1998) Mutational analysis of the complex of human RNase inhibitor and human eosinophil-derived neurotoxin (RNase 2) .Teufel D.P., Kao R.Y., Acharya K.R., Shapiro R.Biochemistry 42:1451-1459(2003) Eosinophil-derived neurotoxin (EDN) , an antimicrobial protein with chemotactic activities for dendritic cells.Yang D., Rosenberg H.F., Chen Q., Dyer K.D., Kurosaka K., Oppenheim J.J.Blood 102:3396-3403(2003) Post-translational tyrosine nitration of eosinophil granule toxins mediated by eosinophil peroxidase.Ulrich M., Petre A., Youhnovski N., Proemm F., Schirle M., Schumm M., Pero R.S., Doyle A., Checkel J., Kita H., Thiyagarajan N., Acharya K.R., Schmid-Grendelmeier P., Simon H.-U., Schwarz H., Tsutsui M., Shimokawa H., Bellon G., Lee J.J., Przybylski M., Doering G.J. Biol. Chem. 283:28629-28640(2008) An enzyme assisted RP-RPLC approach for in-depth analysis of human liver phosphoproteome.Bian Y., Song C., Cheng K., Dong M., Wang F., Huang J., Sun D., Wang L., Ye M., Zou H.J. Proteomics 96:253-262(2014) X-ray crystallographic structure of recombinant eosinophil-derived neurotoxin at 1.83-A resolution.Mosimann S.C., Newton D.L., Youle R.J., James M.N.G.J. Mol. Biol. 260:540-552(1996) Mapping the ribonucleolytic active site of eosinophil-derived neurotoxin (EDN) . High resolution crystal structures of EDN complexes with adenylic nucleotide inhibitors.Leonidas D.D., Boix E., Prill R., Suzuki M., Turton R., Minson K., Swaminathan G.J., Youle R.J., Acharya K.R.J. Biol. Chem. 276:15009-15017(2001) Molecular recognition of human eosinophil-derived neurotoxin (RNase 2) by placental ribonuclease inhibitor.Iyer S., Holloway D.E., Kumar K., Shapiro R., Acharya K.R.J. Mol. Biol. 347:637-655(2005) Crystal structures of eosinophil-derived neurotoxin (EDN) in complex with the inhibitors 5'-ATP, Ap3A, Ap4A, and Ap5A.Baker M.D., Holloway D.E., Swaminathan G.J., Acharya K.R.Biochemistry 45:416-426(2006)
NCBI and Uniprot Product Information
NCBI GI #
NCBI GeneID
NCBI Accession #
NCBI GenBank Nucleotide #
Molecular Weight
19.5 kDa
NCBI Official Full Name
non-secretory ribonuclease
NCBI Official Synonym Full Names
ribonuclease A family member 2
NCBI Official Symbol
RNASE2
NCBI Official Synonym Symbols
EDN; RAF3; RNS2
NCBI Protein Information
non-secretory ribonuclease
UniProt Protein Name
Non-secretory ribonuclease
UniProt Gene Name
RNASE2
UniProt Synonym Gene Names
EDN; RNS2; RNase 2
UniProt Entry Name
RNAS2_HUMAN
Customer Reviews
Loading reviews...
Share Your Experience
Similar Products
Product Notes
The RNASE2 rnase2 (Catalog #AAA114100) is a Recombinant Protein produced from E Coli or Yeast or Baculovirus or Mammalian Cell and is intended for research purposes only. The product is available for immediate purchase. The immunogen sequence is 28-161aa; Full Length of Mature Protein. The amino acid sequence is listed below: KPPQFTWAQW FETQHINMTS QQCTNAMQVI NNYQRRCKNQ NTFLLTTFAN VVNVCGNPNM TCPSNKTRKN CHHSGSQVPL IHCNLTTPSP QNISNCRYAQ TPANMFYIVA CDNRDQRRDP PQYPVVPVHL DRII. It is sometimes possible for the material contained within the vial of "Non-secretory ribonuclease, Recombinant Protein" to become dispersed throughout the inside of the vial, particularly around the seal of said vial, during shipment and storage. We always suggest centrifuging these vials to consolidate all of the liquid away from the lid and to the bottom of the vial prior to opening. Please be advised that certain products may require dry ice for shipping and that, if this is the case, an additional dry ice fee may also be required.Precautions
All products in the AAA Biotech catalog are strictly for research-use only, and are absolutely not suitable for use in any sort of medical, therapeutic, prophylactic, in-vivo, or diagnostic capacity. By purchasing a product from AAA Biotech, you are explicitly certifying that said products will be properly tested and used in line with industry standard. AAA Biotech and its authorized distribution partners reserve the right to refuse to fulfill any order if we have any indication that a purchaser may be intending to use a product outside of our accepted criteria.Disclaimer
Though we do strive to guarantee the information represented in this datasheet, AAA Biotech cannot be held responsible for any oversights or imprecisions. AAA Biotech reserves the right to adjust any aspect of this datasheet at any time and without notice. It is the responsibility of the customer to inform AAA Biotech of any product performance issues observed or experienced within 30 days of receipt of said product. To see additional details on this or any of our other policies, please see our Terms & Conditions page.Item has been added to Shopping Cart
If you are ready to order, navigate to Shopping Cart and get ready to checkout.