In this Article
All of the products listed in AAA Biotech’s catalog are strictly for research-use only (RUO).

Stop second-guessing your assay. Here's a researcher’s step-by-step guide and detailed explanation of factors you need to evaluate - from format and sensitivity to validation data and species compatibility for efficient selection of the right ELISA kit.
Cynthia Lee
Lead Clinical Research Coordinator (LCRC)
Quick Summary
- Match your ELISA kit format (direct, indirect, sandwich, or competitive) to your specific research goal.
- Always verify species compatibility and sample-type validation before purchasing.
- Prioritize ELISA test sensitivity and assay range that fits your expected analyte concentration.
- Review lot-to-lot precision, cross-reactivity data, and the certificate of analysis (COA).
- Confirm all kit components are included and check manufacturer citations and support quality.
The enzyme-linked immunosorbent assay, better known as the ELISA, remains one of the most relied-upon tools in life science research, clinical diagnostics, and drug development. Its ability to detect and quantify proteins, hormones, antibodies, and peptides from complex biological matrices makes it a cornerstone of the modern lab.
But with hundreds of ELISA testing kits available across vendors, picking the wrong one wastes reagents, time, and precious samples. A mismatched kit can produce false positives, poor sensitivity, or simply fail to detect your target at all.
This guide walks you through every critical decision point — so you arrive at the right ELISA kit for research from the very first run.
Step 1: Define the Purpose of Your ELISA Assay
Before browsing any catalog, a researcher must answer these three questions:
- What are you measuring? A cytokine, hormone, antibody titer, or small molecule?
- Are you quantifying or qualifying? Absolute concentrations vs. presence/absence changes the kit requirements.
- Is this for discovery research, validation, or ELISA diagnostic kits for clinical use? Regulatory-grade applications demand stricter validation standards.
Understanding the purpose of your ELISA assay upfront prevents expensive mistakes later. Many researchers skip this step and discover mid-experiment that their kit lacks the sensitivity range they need.
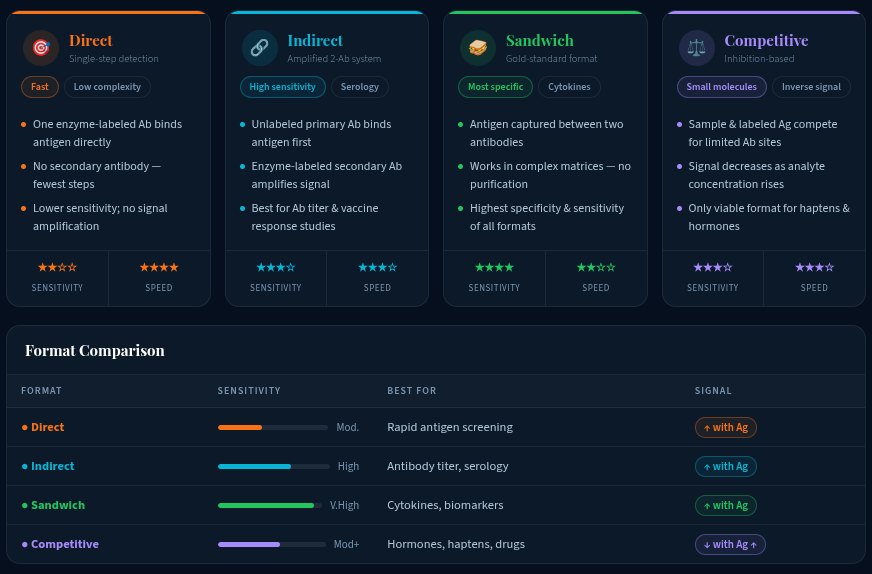
Step 2: Select the Right Type of ELISA for Your Application
The four main types of ELISA differ in how the antigen–antibody complex is captured and detected. Choosing the wrong format is one of the most common and costly errors in assay setup.
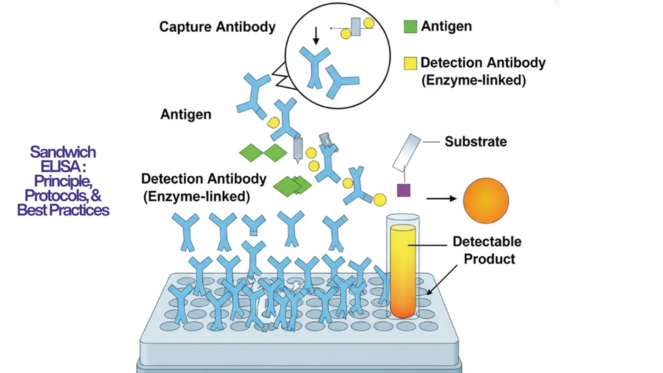
For most biomarker-focused ELISA-based assay work, sandwich ELISA is the preferred choice due to its superior specificity and sensitivity. However, if you're working with a small-molecule drug metabolite or a hapten, competitive ELISA is the only viable approach.
Step 3: Confirm Species Reactivity and Sample Compatibility
Many kit failures trace back to a single overlooked detail: the kit was never validated for your species or sample type.
- Common species: Human, mouse, and rat kits are widely available with robust validation data.
- Cross-species use: If your target protein shows ≥ 90% sequence homology across species, a human-validated kit may work in other models — but always confirm with the manufacturer whether or not this is the case with a given kit.
- Sample matrices matter: Results from EDTA plasma, heparin plasma, serum, urine, and cell culture supernatant are not interchangeable. Each matrix can introduce interference affecting your signal.
💡Pro Tip: If you are using a non-standard matrix (e.g., synovial fluid, CSF, or tissue lysate), look specifically for kits that include dilution linearity data in that matrix — not just serum/plasma.
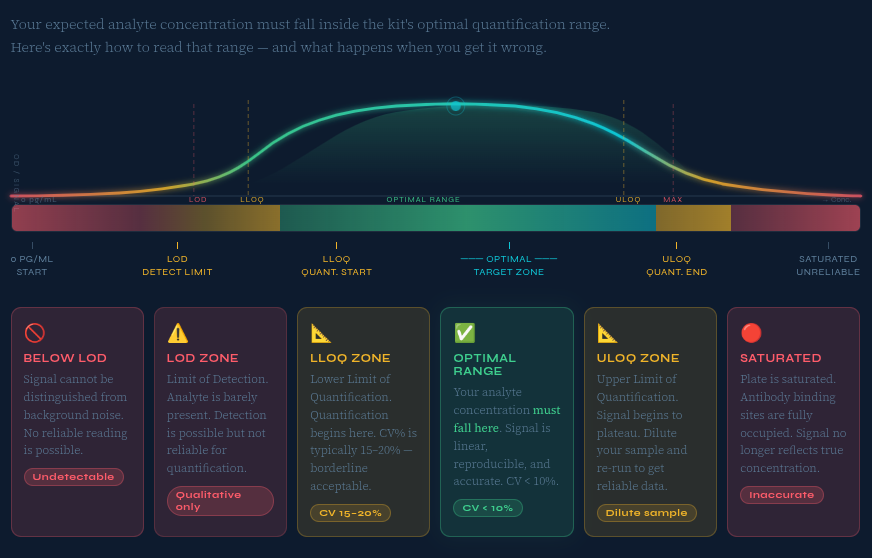
Step 4: Match ELISA Test Sensitivity to Your Analyte Concentration
ELISA test sensitivity is arguably the most misunderstood selection parameter. Two key metrics define it:
- Limit of Detection (LOD): The lowest concentration the assay can reliably distinguish from background noise.
- Lower Limit of Quantification (LLOQ): The lowest concentration that can be measured with acceptable precision (CV ≤ 20%).
⚠️ Common mistake: Choosing the most sensitive ELISA kit available regardless of your sample's actual concentration. High-sensitivity kits have a narrower dynamic range, meaning concentrated samples must be diluted multiple times — adding variability and error.
Step 5: Scrutinize the Kit's Validation and Performance Data
A well-validated ELISA kit for research should have documented data covering all of the following parameters. If these are missing from the datasheet, ask the manufacturer directly — or look elsewhere.
| Validation Parameter | What to Look For | Acceptable Benchmark |
|---|---|---|
| Intra-assay Precision (CV%) | Variation within a single plate run | CV < 10% |
| Inter-assay Precision (CV%) | Variation across different lots/runs | CV < 15% |
| Spike Recovery / Accuracy | % recovery of a known amount of spiked analyte | 80–120% |
| Dilution Linearity / Parallelism | Signal drops proportionally as the sample is diluted | R² ≥ 0.99 |
| Cross-Reactivity | False signal from structurally similar proteins | < 5% |
| Freeze-Thaw Stability | Analyte integrity after repeated freeze-thaw | ≥ 3 cycles documented |
Step 6: Check Kit Completeness, Cost, and Manufacturer Credibility
KIT COMPONENTS CHECKLIST
- Pre-coated 96-well microplate (preferably with removable strip wells)
- Recombinant standard protein in a quantified lyophilized or liquid form
- Assay/sample diluent buffer
- Detection antibody (conjugated or unconjugated, depending on format)
- Substrate solution (e.g., TMB) and stop solution
- Positive and negative controls with expected ranges
- Wash buffer concentrate
MANUFACTURER CREDIBILITY
- Original manufacturer vs. reseller: Many ELISA testing kits are rebranded from a single source. Buying directly from the original manufacturer ensures proper technical support and access to supplementary reagents.
- Publication record: Search PubMed for studies citing the specific catalog number. Peer-reviewed citations are the strongest evidence of real-world performance.
- Responsive support: Technical teams that can provide additional calibrators, troubleshoot matrix effects, or share detailed validation SOPs are invaluable — especially when using ELISA diagnostic kits in regulated environments.
Quick-Reference Decision Table
| Your Situation | Recommended Priority |
|---|---|
| Measuring pg/mL cytokines in CSF or urine | High-sensitivity kit; confirm LLOQ in that specific matrix |
| Working with a non-human model (rat, rabbit, pig) | Check species-specific or cross-reactive validated kit |
| Running high-throughput drug screening | 96-well sandwich ELISA with removable strip format |
| Regulatory/clinical diagnostic use | ISO-certified ELISA diagnostic kits with GMP-grade validation |
| First-time ELISA for a novel target | Choose a broad dynamic range kit; run a spiked recovery test first |
Conclusion
Choosing the right ELISA kit is not guesswork - it's a structured decision. When you align your format, sensitivity, species validation, and performance data with your specific research goal, you eliminate wasted runs and unreliable results.
Take the time to evaluate each parameter carefully, and your assay will deliver the reproducible, publication-ready data your research deserves.
Faq's
What is the difference between direct, indirect, sandwich, and competitive ELISA?
These are the four main types of ELISA, each differing in how the target antigen is captured and detected. Direct ELISA uses a single labeled antibody and is the fastest. Indirect ELISA uses a secondary antibody for signal amplification, making it more sensitive. Sandwich ELISA uses two antibodies (capture + detection) and offers the highest specificity for complex samples. Competitive ELISA is used for small molecules or haptens where only one antibody binding site is available.
How do I know if an ELISA kit is sensitive enough for my samples?
Review the kit's Lower Limit of Quantification (LLOQ) and confirm it falls below the lowest concentration you expect in your samples. For very low-abundance analytes (e.g., in CSF or urine), choose high-sensitivity kits rated in the single-digit pg/mL range. Also, check that published reference values for your analyte fall comfortably within the kit's working range.
Can I use a human ELISA kit for mouse or rat samples?
Sometimes, but generally only recommended if the target protein shows ≥ 90% amino acid sequence homology between species. Some manufacturers explicitly document cross-species reactivity in their datasheets. Always contact the manufacturer before substituting species, as even minor structural differences at the epitope can prevent antibody binding and produce false-negative results.
What CV% should I look for when evaluating ELISA kit precision?
For intra-assay (within-run) precision, a CV% below 10% is considered acceptable for most research applications. For inter-assay (run-to-run) precision, a CV% below 15% is the standard benchmark. Anything above these thresholds suggests poor reproducibility, which will undermine your ability to compare results across experiments or time points.
What is the purpose of a sandwich ELISA vs. a competitive ELISA?
The purpose of a sandwich ELISA is to detect and quantify large proteins (like cytokines, growth factors, or immunoglobulins) in complex matrices with high specificity and sensitivity. A competitive ELISA serves a different purpose: it is designed for small molecules — such as steroid hormones, drugs, or peptides, that are too small to simultaneously bind two antibodies. The signal in competitive ELISA inversely correlates with analyte concentration.
References / Studies Used
01. Hayrapetyan H, Tran T, Tellez-Corrales E, Madiraju C. Enzyme-Linked Immunosorbent Assay: Types and Applications. Methods Mol Biol. 2023;2612:1–17. PubMed
02. Lequin RM. Enzyme Immunoassay (EIA)/Enzyme-Linked Immunosorbent Assay (ELISA). Clin Chem. 2005;51(12):2415–2418. Oxford Academic
03. Biomedica Medizinprodukte. How to Choose the Right ELISA Kit. 2024. bmgrp.com
04. Boster Biological Technology. How to Choose a Good ELISA Kit. 2023. bosterbio.com
05. Engvall E, Perlmann P. Enzyme-Linked Immunosorbent Assay (ELISA): Quantitative Assay of Immunoglobulin G. Immunochemistry. 1971;8(9):871–874.
Cynthia Lee
Lead Clinical Research Coordinator (LCRC)
Cynthia Lee specializes in understanding highly validated and characterized monoclonal/polyclonal antibodies, recombinant proteins, and ELISA kits. Her study primarily focuses on immunoassay optimization and biomarker validation across oncology and immunology research programs.

 Cynthia
Cynthia